Community-led intervention curbs childhood obesity and boosts quality of life
Team Metabolic Health
A new study finds that empowering communities with systems-based tools to fight childhood obesity not only reduced weight trends but also improved quality of life in boys—suggesting a resilient, community-first approach to lasting health changes.
The RESPOND randomized trial is a community-based systems intervention to prevent childhood obesity and non-communicable diseases.
Scientists at Deakin University recently evaluated the trial’s three-year behavioral, health-related quality of life, and body mass index outcomes. The study is published in the journal Public Health.
Background
Childhood obesity is a serious public health concern as it often persists into adulthood and increases the risk of several non-communicable diseases.
The complex and multifactorial nature of childhood obesity risk factors may lead to resistance to intervention. This complexity includes multiple factors that can influence a child’s food consumption, including early-life exposure to a variety of foods, food availability at home and within the community, and socioeconomic status.
Existing evidence indicates that multi-level interventions that apply multiple strategies across communities can significantly reduce children’s body weight.
Systems science and systems thinking are methods of understanding the relationships and connections between component factors driving complex systems. Thus, they can provide promising and innovative interventions to prevent childhood obesity.
The Reflexive Evidence and Systems Interventions to Prevent Obesity and Non-communicable Disease (RESPOND) trial was initiated to examine whether building the capacity of key community health staff at scale through systems-based training can develop a sustainable model for delivering such interventions for childhood obesity prevention.
In this study, scientists have evaluated the impact of the RESPOND trial on BMI z-scores (standardized body mass index), health-related quality of life, and related behaviors.

Study: Three-year behavioural, health-related quality of life, and body mass index outcomes from the RESPOND randomized trial. Image Credit: chanchai plongern/Shutterstock
Study Design
RESPOND was designed as a four-year randomized childhood obesity prevention trial involving 10 local government areas in northeast Victoria, Australia. The aim was to build community capacity to apply systems science to prevent childhood obesity and non-communicable diseases.
The intervention was initially implemented in the selected areas in July 2019 (step one) or July 2021 (step two). Baseline child-level data were collected between March and June 2019, and the follow-up data collection was planned for 2021 and 2023.
Due to the COVID-19 pandemic, however, step two communities could not begin their intervention as planned and were therefore treated as controls.
The step one communities partially implemented the intervention actions and were defined as “intervention communities.” The trial was altered to a cluster-parallel randomized design, allowing the researchers to compare outcomes in intervention and control communities.
In this study, scientists compared intervention versus control communities over three years (2019–2022). The study analyzed data obtained from 31 primary schools that participated in both March to June 2019 and March to August 2022 data collection waves.
The three-year outcomes included BMI z-scores, health-related quality of life, and self-reported health behaviors.
Important Observations
The analysis conducted within intervention communities revealed a small reduction in BMI z-scores and the percentage of overweight or obesity from 2019 to 2022. Within control communities, a small increase in BMI z-scores and the percentage of overweight or obesity was observed during the same period.
Self-reported health behaviors revealed limited effects of the intervention on any analyzed outcomes (physical activity, recreational screen time, and frequency of takeaway food consumption). However, a noteworthy intervention effect on daily water consumption was observed in boys.
Between 2019 and 2022, overall health-related quality of life deteriorated for children, likely due to COVID-19-related challenges. However, for boys, a significant improvement in quality of life and psychosocial health was observed within intervention communities.
Study Significance
The study finds a limited but positive effect of the community-based systems intervention (RESPOND) on psychosocial health, overall quality of life, and water consumption among boys who participated in the trial. However, the intervention had no significant impact on these metrics for girls.
The study also finds a general decline in several obesity-related behaviors among boys and girls in both intervention and control communities over the study period, which researchers attribute in part to the pandemic’s impact on lifestyle behaviors.
A non-significant but favorable effect of the intervention has been observed for BMI z-scores, overweight or obesity prevalence, and several other behavioral outcomes (active transport to and from school, sleep, and consumption of takeaway food and sweetened drinks).
The RESPOND trial utilized systems thinking to help communities identify and target local childhood obesity risk factors. The researchers emphasize that understanding these local factors is crucial in assessing the intervention’s overall merit.
The findings suggest that empowering communities to use systems thinking in obesity prevention could have a protective effect on children’s health, even in the face of major challenges like the COVID-19 pandemic.
Given the scarcity of effective interventions to address childhood obesity and non-communicable diseases, building capacity within communities to deliver systems-based interventions seems to be a promising approach.
Further analyses and investments in RESPOND’s findings could lead to broader training of community health workers in systems thinking and enable the creation of a learning system for effective and rapid knowledge acquisition. Further investigation is also needed into the observed gender differences in intervention effects.
Credit: news-medical.net
Can millets raise blood sugar levels too: A new study tells you when
Team Metabolic Health
Smart guide on which variety to look out for and how to have it
Have you been buying packaged millets from shop shelves thinking you are making a healthy choice? Chances are they are polished millets which could be spiking your sugar levels just like any other carbohydrate. While polished millets bring down the cooking time and ease digestion, they also lead to the loss of minerals and phytochemicals that make them healthy. Debranning — or removing the outer layers of the grain — also makes them starchy and high in glycemic index, according to Dr Shobana Shanmugam, head of the department of diabetes food technology at Madras Diabetes Research Foundation.

Millets cooked like rice are preferable to chapatis made of millets as they hold more moisture and are easier to eat. (File)
“Integrating different millets into the diet — instead of wheat and rice — can help in increasing food diversity. This is essential for combating the triple burden of malnutrition in India — undernutrition, overnutrition, and micronutrient deficiency. But debranning reduces food value,” she says. Her team recently published a paper in the journal Springer Nature on the impact of debranning on five Indian millets — foxtail, little, kodo, barnyard, and proso.
What happens when millets are debranned or polished?
Debranning leads to loss of minerals — iron, calcium, zinc, phosphorus, magnesium — in all five millets. “In fact, iron levels went below the level of detection in the grains in our studies,” says Dr Shanmugam. The process also led to the loss of protein content in all but two of the millets — little and barnyard millets. These are likely to contain the protein in the inner layers.
Significantly, the available carbohydrate went up across all five millets when polished by up to 11 gm per 100 gm of the millet. “We looked at the grains with electron microscopy to check which are the layers that have been removed. And, higher the degree of polish, the more starchy the grain becomes,” says Dr Shanmugam.
“While there is awareness that millets are better for your health, people might not know that these grains too can be polished. And, that the process can remove a lot of the nutrients. Yet, many polished millets make health claims that can be misleading,” she warns.
How can one tell whether the millet has been polished?
Dr Shanmugam suggests a few ways one can tell whether a grain has been polished or not:
1) Polished grains look whiter than the unpolished ones, which are dull.
2) Two, the grains should be glassy because of the oil content in the outer layers.
3) The polished grains are likely to be smoother to touch while the unpolished ones are rougher.
How can millets be integrated into our diets?
Millets cooked like rice are preferable to chapatis made of millets as they hold more moisture and are easier to eat. “If you see rice, it expands quite a bit when cooked. Around 70 per cent of the rice we eat is water; chapatis are the opposite and will have about 30 per cent water.”
Bottom of Form
She also warns against diabetics eating porridge made of millets. “When we make millet porridge, it again increases the glycemic index and can raise blood glucose levels.”
Credit: The Indian Express
Weight loss injections and cancer: 6 questions, answered
Team Metabolic Health
Most people know that losing weight has numerous benefits, but making major lifestyle changes can be extremely difficult. For example, obese people are often at risk for injuries when they start a new exercise routine, which can set them back or ultimately cause them to give up on it. As a result, more and more people have been turning to weight loss medications for help.
In the last couple of years, a new class of weight-loss injection drugs called GLP-1 agonists like semaglutide, tirzepatide, liraglutide and dulaglutide has made headlines for their promise to help people lose weight. The drugs go by several different brand names, and more are being approved all the time. But what are they, how do they work and are there things current cancer patients should be aware of if they’re considering these drugs?
For answers to these questions, we talked to endocrine neoplasia and hormonal disorders specialist Sonali Thosani, M.D.
How do these drugs work, and how are they different from previous weight loss drugs?
Previous weight loss drugs worked in a few different ways. Some suppressed appetite and made you feel fuller longer, but these could only be used for a limited time due to the side effects. Others inhibited fat absorption, but were hard to tolerate for many patients because they caused gastrointestinal issues.
This new class of drugs, the GLP-1 agonists, has been used for diabetes treatment since 2005, but they only have gotten approval for obesity treatment in the last decade. They work by slowing down gastric emptying. This makes patients feel fuller longer, helping them eat less. They also help balance blood sugars after eating by stimulating insulin secretion and inhibiting glucagon secretion.
Studies have shown that patients can lose up to 20% of their body weight with GLP-1 agonists. That makes these drugs comparable to surgical options, like gastric sleeve surgeries. However, they also have some side effects, with many patients experiencing nausea, vomiting and diarrhea. There have also been some recent reports that they might be linked to gastroparesis in some patients. This is a paralysis of the stomach that can be debilitating and hard to treat.
What’s also interesting about these drugs is that GLP-1 is a neuropeptide. This means it’s made in the neuronal cells in the brain and released in the hypothalamus, which controls several metabolic and cardiovascular functions. So, it will be interesting to see what we continue to learn about how these drugs work and what the long-term effects might be on other organ systems.
Can weight loss injections decrease your cancer risk?
This is a more complicated question than it seems because obesity is so closely associated with increased cancer risk. There are really two questions here:
Do the drugs themselves decrease cancer risk?
Does their ability to decrease obesity result in a lowered cancer risk?
There is significant evidence that the answer to the second question is yes, with the caveat that patients should know that these drugs aren’t meant to be taken forever. Many people take GLP-1 agonists or other weight loss medication, they lose weight, they go off the drugs and then they gain the weight back. That’s not going to help lower long-term cancer risk.
Where these types of drugs can be helpful is that many obese people have a hard time making healthy lifestyle changes because it’s very difficult for them to exercise. If taking these drugs for a limited amount of time can help some lose weight quickly so they can get on an exercise plan they can stick to, then that’s helping lower cancer risk long-term. But it all comes back to the person making and sticking to those lifestyle changes, even after they stop taking the drugs. If they can do that, then you’re probably seeing a net positive about long-term cancer risk or other long-term health risks.
As for whether the drugs themselves reduce cancer risk, it’s too early to say for sure. But there is some evidence that GLP-1 agonists like semaglutide may decrease cancer risk beyond just their ability to help people lose weight. A study published early this year showed that patients with diabetes who used weight loss injections were less likely to have colorectal cancer compared to patients on other diabetes drugs, regardless of whether the patients were overweight.
Are there concerns that weight loss injections could increase cancer risk?
There was an early study a couple of years ago that indicated a possible link to thyroid cancer, but that study had some limitations, and subsequent studies have shown that there is likely not a link there. The European Medical Agency even announced that they did not find a causal link after a months-long review of the data.
There has also been some concern that some of the side effects could increase the risk of pancreatic cancer, but so far, studies have not shown that to be the case.
If cancer isn’t a major concern, are there other concerns about these drugs?
There’s always a bit of wariness when a new class of drugs comes to market because we’ve seen several times where promising drugs came to market for weight loss and then they’ve had to be pulled back, due to safety concerns. What’s unique about GLP-1 agonists compared to other weight loss drugs is that we already have a lot of experience with them in patients with diabetes. We also know from the published data that these drugs have heart health benefits; this sets them apart from other weight loss drugs currently on the market.
The primary concern is the availability of these drugs for the patients who need them. They’re expensive, and it’s been hard to get insurers to cover them, even when a patient has a clear indication for them. And there are celebrities and influencers using them who don’t have indications for their use, adding to a national shortage and making them even harder to get.
And the gastroparesis issue is not a small thing. Doctors need to be selective when deciding whether a patient meets the criteria for these drugs because gastroparesis can be very difficult to manage. Guidelines from anesthesia and surgical societies recommend holding the weekly formulations of these drugs for at least a week before surgery, so careful planning is required if a patient is using these therapies.
Are there concerns about these drugs for people who have already had cancer?
There are a couple of things to note for cancer patients, which is why it is always very important to discuss these things with your care team.
We generally avoid GLP-1 agonists for pancreatic cancer patients, for example, because these patients are already at high risk for pancreatitis, and these drugs do carry some potential risk for that. We are also cautious about using these drugs if you’re on active cancer therapy and experiencing side effects such as nausea and vomiting; GLP-1 agonist therapy can sometimes worsen those.
These drugs cause dramatic weight loss, some of which is also a significant muscle mass loss. So, we need to carefully monitor their use in patients who are on active chemotherapy, as sarcopenia and weight loss can make it harder for patients to tolerate chemotherapy.
Are there alternatives to weight loss drugs that people should consider?
I think bariatric surgery is underutilized. And, of the 20 million adults in the US who suffer from severe obesity, less than 1% will undergo bariatric surgery annually. There was a study presented at a national meeting looking at the role of bariatric surgery in patients with obesity and the impact on cancer prevention, and it found a marked decrease in cancer diagnoses in patients with obesity who underwent bariatric surgery compared to those who didn’t. This was really the first study to address this question, and I think this exciting new area will have more studies coming out in the next 10 to 15 years.
Credit: mdanderson.org
Limiting fast-food outlets can reduce childhood overweight and obesity
Team Metabolic Health
Restricting new fast-food sellers in areas where there are already a large number could reduce obesity and overweight in children.
In a recent study published in obesity, researchers investigated whether a policy in England that banned new fast-food outlets effectively reduced obesity and overweight in children.
Their findings indicated that restricting new fast-food sellers in areas where there are already a large number could reduce obesity and overweight in children.

Study: Planning policies to restrict fast food and inequalities in child weight in England: a quasi-experimental analysis. Image Credit: Paulo PFZ STUDIO/Shutterstock.com
Background
The United Kingdom has among the highest rates of childhood obesity in Europe, with obesity among ten and 11-year-olds increasing from slightly under 32% in 2006-07 to nearly 41% in 2020-21. The pandemic is thought to have been one of the drivers for this change.
Childhood obesity is associated with several short-term and long-term effects. In the short term, it reduces life quality and is linked to depression, anxiety, stigma, bullying, and self-esteem issues.
In the long term, childhood obesity can continue into adulthood and increase the risk of mental disorders, diabetes, and cardiovascular diseases. The cost of obesity-related conditions for the National Health Service was estimated to be £6.1 million in 2015, and this is expected to rise to £9.7 billion by 2050.
Complex factors influence obesity, but environmental factors, especially outside the home, play a major role. Fast food intake and the density of fast-food outlets have been linked to higher energy consumption and obesity, particularly in low-income areas.
The density of fast-food outlets in England has increased from 142 per 100,000 residents in 2019 to 170 in 2021. The impacts are unequally felt, as deprived areas contain five times more outlets than wealthier areas.
In recent years, half of local governments have introduced planning guidelines that target limits on new outlets and promote healthier food environments. Gateshead, among the most deprived areas, implemented stricter planning policies to reduce childhood obesity.
A complete ban on new fast-food outlets since 2015 aimed to lower obesity in 10 and 11-year-olds from 23% to less than 10% by 2025. Studies show a reduction in fast-food outlets in Gateshead compared to areas without similar policies.
About the Study
In this study, researchers investigated whether reducing fast-food outlets leads to changes in childhood obesity and reduces health inequalities, exploring the impact of the policy within five years and whether it is more effective in deprived areas with high fast-food outlet density.
They gathered data on children’s weight and food outlets from national monitoring programs, also including population size and area-level deprivation indices, with higher scores indicating more deprivation.
The outcome variable was the prevalence of obesity and overweight in 10 and 11-year-old children, while outlet density was calculated as the number of outlets for every 100,000 residents in each area.
For the analysis, researchers considered areas in Gateshead to be the treatment group and matched them to areas from nearby regions that did not implement similar restrictions.
They then used statistical models to analyze the impact of restricting fast-food outlets, controlling for deprivation scores.
Findings
Areas in Gateshead had a consistently higher prevalence of obesity and overweight among children aged 10 and 11 compared to the control areas. Gateshead also had a higher density of fast-food outlets compared to control areas throughout the years.
The prevalence decreased from 38% in 2011 to 35.5% in 2015 but rose again to 37.7% by 2020. Over time, the difference in prevalence between Gateshead and the control areas narrowed.
The impact of planning guidance on reducing obesity and overweight prevalence was not statistically significant overall, but area-level deprivation scores did show a strong effect.
Notably, the planning policy had a significant reduction in obesity and overweight prevalence in the second and third-most deprived areas. This effect remained for the second most deprived area after sensitivity analysis.
Conclusions
The study provides evidence of the effectiveness of local planning policy in reducing obesity and overweight in children aged 10 and 11 within four years of implementation. There was a 10% reduction in fast-food outlet density in Gateshead within four years of the policy.
Decreases in prevalence were observed in the second most deprived areas, which also had the highest concentration of fast-food outlets before the policy.
Researchers found no effects at the population level and noted that the effect could take longer than four years to be measurable.
These findings have policy implications, indicating that planning policies could help reduce inequalities in childhood obesity and overweight, relying on a structural approach to improve health outcomes rather than relying on changes in individual behavior.
However, the growing presence of online food delivery needs further research to understand its impact on access to unhealthy food and whether current policies need updates to address the changing food environment.
Credit: news-medical.net
Perks vs Side-Effects Of Ozempic: What’s The Real Story Behind This GLP-1 Drug?
Team Metabolic Health
Ozempic, prescribed for adults with type 2 diabetes, comes with serious drawbacks, while being valued for its benefits. Find out if Ozempic is worth it?

Ozempic | Image: iStock
Usually used to lower blood sugar levels in individuals with type-2 diabetes, Ozempic is back on the lips of medical experts after a recent study revealed the possibility of semaglutide, the active ingredient in Ozempic and Wegovy, proving effective in reducing the risk of Alzheimer’s in patients with Type 2 diabetes.
These recent revelations published in Alzheimer’s & Dementia support the nuero-protective benefits linked with GLP-1 receptors agonists. Ozempic has also been utilised to combat cardiovascular issues such as stroke and heart attack in people suffering from type 2 diabetes.
Benefits of Ozempic
The presence of GLP-1 receptors in the brain suggests that activating these receptors may offer neuroprotective effects, potentially aiding in the treatment of neurodegenerative diseases such as Parkinson’s and Alzheimer’s. A study in Nature noted that GLP-1 agonists can decrease inflammation in brain tissue, which is crucial since chronic inflammation is linked to cognitive decline.
Stable blood sugar levels are vital for mental well-being. Fluctuations in blood glucose can lead to mood disturbances, fatigue, and irritability, increasing the risk of anxiety and depression. By mitigating these spikes, Ozempic may indirectly enhance mood and cognitive clarity.
Credit: Republic World
Can a popular weight-loss drug reduce risk of Alzheimer’s?
Team Metabolic Health
Alzheimer’s causes a gradual decline in memory, thinking, behaviour, and social skills, and it is the most common cause of dementia.
According to the new study published in the journal Alzheimer’s & Dementia on Thursday, semaglutide was found to significantly lower the risk of developing AD compared to other diabetes medicines. Specifically, it was found to reduce the risk of a first-time Alzheimer’s diagnosis by 40% to 70%.
Semaglutide, the active ingredient in popular blood sugar control and weight loss drugs such as Ozempic and Wegovy, can reduce the risk of Alzheimer’s disease (AD) in people with type 2 diabetes, a new study shows.
Alzheimer’s causes a gradual decline in memory, thinking, behaviour, and social skills, and it is the most common cause of dementia. According to the new study published in the journal Alzheimer’s & Dementia on Thursday, semaglutide was found to significantly lower the risk of developing AD compared to other diabetes medicines. Specifically, it was found to reduce the risk of a first-time Alzheimer’s diagnosis by 40% to 70%.
“These results were similar for older patients, both genders, and those with and without obesity. Cumulative incidence curves [of semaglutide and other diabetes medicine] began to diverge within 30 days, and continued to separate thereafter, indicating semaglutide’s potential to delay or slow Alzheimer’s Disease development with sustained effects,” the study said.

A promising study
Researchers analysed health records of over a million patients in the US with type 2 diabetes to see how semaglutide affects the risk of developing AD. They compared semaglutide to seven other diabetes medications, and tracked the patients for up to three years to see if they were diagnosed with Alzheimer’s. The other diabetes drugs used in the study included metformin, insulin, and older GLP-1 drugs like liraglutide.
Using specific statistical methods, they studied the time it took for Alzheimer’s to be diagnosed in these patients, if at all. The study found that patients prescribed semaglutide had a significantly lower risk of AD than those who had taken one of the seven other diabetes drugs.
The most notable difference with regards to AD incidence was seen between patients who took semaglutide and those who took insulin, with semaglutide patients boasting a 70% lower risk of developing Alzheimer’s.
Semaglutide also outperformed its predecessor liraglutide. The study’s senior author Rong Xu, the director of the Center for Artificial Intelligence in Drug Discovery at Case Western Reserve University School of Medicine in Ohio, speculated based on this finding that more potent GLP-1 RA drugs might have an even stronger effect in combating Alzheimer’s.
“If there is a higher dose form, are we going to see an even stronger effect?” Xu said in an interview to NBC.
Science behind findings
The study said GLP-1 RAs, a group of drug which includes semaglutide, might help protect brain function. Early research on semaglutide identifies a number of ways in which it might benefit the brain such as lowering toxic effects of certain proteins linked to AD, improving how brain cells use glucose for energy, and reducing the buildup of harmful plaques and tangles associated with Alzheimer’s.
“Semaglutide reduces neuro-inflammation, which is commonly linked to Alzheimer’s. It helps stabilise blood sugar levels, limiting oxidative stress or cell damage and enhancing cellular energy. This action can protect neurons from degeneration, a hallmark of Alzheimer’s and other neurodegenerative diseases,” Dr (Prof) M V Padma Srivastava, chairperson, Neurology, Paras Health, Gurugram, told The Indian Express.
“The study adds to our understanding of how blood glucose control via GLP-1 may offer brain-protective effects…[and] shows that effective blood glucose management and inflammation control are critical in Alzheimer’s progression,” she said.
Potential implications
The US Food and Drug Administration (FDA) has approved two treatments — Biogen’s Leqembi and Eli Lilly’s Kisunla — that marginally slow the progression of AD by targeting the disease’s hallmark amyloid plaques in the brain. But these can cause serious side effects, including brain swelling and brain bleeding. Traditionally, Alzheimer’s has been managed largely using cognitive and lifestyle interventions.
This is what makes the latest research, which introduces a new therapeutic pathway for treating Alzheimer, potentially ground-breaking. “By demonstrating that GLP-1 drugs can mitigate some risk factors for Alzheimer’s, the study suggests a dual-purpose use for these drugs: managing diabetes and offering a preventive mechanism for dementia,” Dr Srivastava said.
For India, this is particularly significant. The country boasts among the highest global rates of type 2 diabetes, which is associated with a higher risk of cognitive decline. This is because type 2 diabetes results in chronic insulin resistance, higher levels of inflammation, oxidative stress in the brain — all of which lead to impaired glucose metabolism in the brain, resulting in cognitive decline and neuron damage over time.
Bottom of Form
“GLP-1 drugs can delay or reduce dementia symptoms in diabetic patients, thus offering a preventive strategy where few currently exist,” according to Dr Srivastava. “Since Alzheimer’s rates are also rising among India’s ageing population, this research could be transformative,” she said.
Credit: The Indian Express
Why more doctors are treating children and adolescents with weight loss drugs
Team Metabolic Health
Watch This Video
Credit: PBS News
How ‘miracle’ weight-loss drugs will change the world
Team Metabolic Health
Models suggest societal upheaval from anti-obesity medicines — but impacts are hard to predict.
Welcome to the healthier, happier world of 2030. Heart attacks and strokes are down 20%. A drop in food consumption has left more money in people’s wallets. Lighter passengers are saving airlines 100 million litres of fuel each year. And billions of people are enjoying a better quality of life, with improvements to their mental and physical health.
These are just some of the ways in which analysts forecast that the new wave of incredibly effective weight-loss drugs, known as GLP-1 agonists, might transform societies and save countries trillions of dollars in the long run. The best known is semaglutide, marketed as Ozempic for diabetes, and as Wegovy for weight loss. “Short of some crazy unfortunate side effect, this is going to change the world,” says Chin Hur, a gastroenterologist at Columbia University in New York City.

Illustration: Karol Banach
It might have already started. In the United States, where 12% of adults say that they have at some stage taken GLP-1 agonists for diabetes or weight loss (see ‘Uptake of GLP-1 drugs in the United States’), media reports suggest that obesity rates are falling, although scientists caution that the data are not statistically significant (see ‘US obesity rates’). Slowing or reversing obesity trends more widely — more than half of the world’s population is expected to be overweight or have obesity by 2035 — would have myriad ripple effects. “The spillover impacts of obesity are enormous,” says Alison Sexton Ward, an economist at the University of Southern California in Los Angeles.
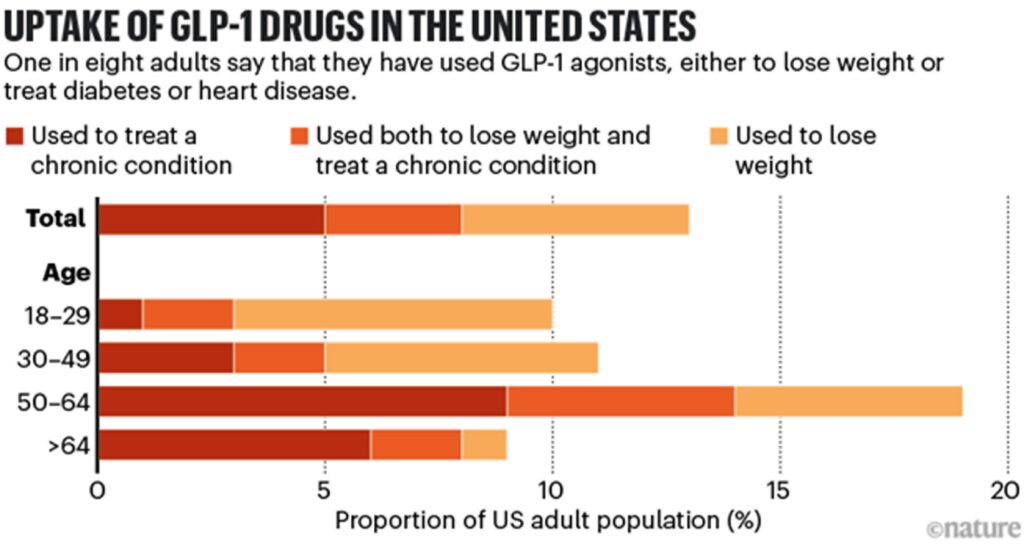
Source: KFF & US NCHS/CDC
But although scientists agree that the drugs could have huge impacts, there is a lot of uncertainty. Efforts to model the weight-loss drugs’ future impact are highly speculative for various reasons, ranging from their high costs to their long-term biological effects, and the big unknown of how people’s behaviour will change. All that has medical researchers and companies scrambling to gather more data and develop better tools to assess how weight-loss drugs might transform societies.
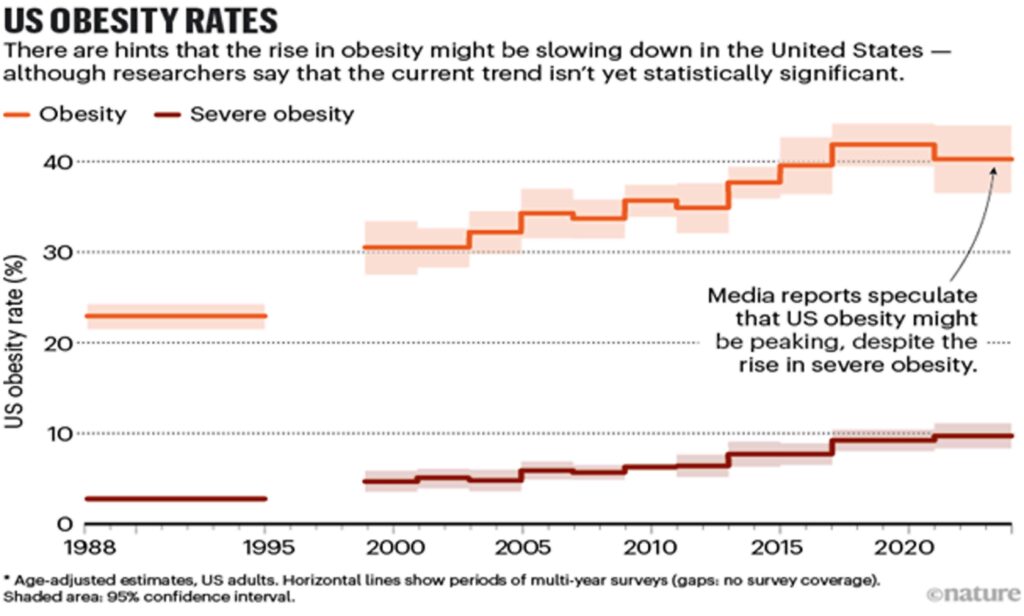
Source: KFF & US NCHS/CDC
Modelling a miracle intervention
Modelling obesity — and its prevention — has long been a staple of public-health research. One way of doing so is to create algorithms that simulate interventions, such as a tax on sugary drinks, or mandatory exercise programmes in schools. By shifting variables such as people’s willingness to cooperate or societal demographics, such models can estimate the health problems that would be prevented or the money that would be saved.
Such policy-based behavioural interventions usually have little effect on preventing weight gain or causing weight loss in the real world, at least in the short term. But the GLP-1 drugs could be different, says Theo Vos, an epidemiologist at the Institute for Health Metrics and Evaluation (IHME) at the University of Washington in Seattle. Trials show that drugs such as semaglutide allow people to lose around 15% of their body weight in 16 months. Next-generation drugs could be even more effective. “This really works, and it works in quite a dramatic way,” says Vos.
The easiest things to model are the immediate impacts of a drug for an individual, including improvements both for physical ailments, such as reduced sleep apnoea, heartburn or joint pain, and in mental health, such as experiencing less social stigma. “If you lose weight, your quality of life improves immediately,” Hur says. Clinical trials suggest that the drugs seem to treat a host of other conditions, too, including addiction, Parkinson’s disease and infertility, but Hur says that it’s still too early to incorporate these benefits into models.
Why do obesity drugs seem to treat so many other ailments?
GLP-1 agonists do have downsides: many users experience nausea, gastrointestinal problems or muscle atrophy, and when people stop taking the drugs, they tend to quickly put the weight back on. Still, GLP-1s are probably different from previous ‘miracle’ weight-loss drugs such as fen–phen (fenfluramine/phentermine), which was banned because of its severe side effects, says Nicolas Rasmussen, a science historian at the University of New South Wales in Sydney, Australia. GLP-1s have been used to treat diabetes for many years and seem to be safe for most people.
Nevertheless, Rasmussen says, “history does tell us they’ll be overused”. Although the drugs might be clearly beneficial for people whose obesity is harming their health, people who take them simply to lose a few kilograms must set that benefit against the side effects. That hasn’t seemed to slow demand, especially as social-media influencers and celebrities promote the drugs for weight loss. Pharmaceutical companies that produce some of the key drugs have experienced shortages.
Economic forecasts
Beyond the effects on individuals, researchers expect that wide use of GLP-1 drugs could have some startling broader economic impacts. Analysts project that a global market already worth US$47 billion this year will grow ten-fold by 2032. People in certain industries, such as the food sector, are already becoming worried by the popularity of the drugs. An analysis by US investment firm Morgan Stanley predicted that US calorie consumption could drop by 1.3% by 2035. Last October, John Furner, the chief executive of retail company Walmart U.S., said that the company had seen a drop in food sales that it attributed to weight-loss drugs. Some analysts have projected less-obvious effects. US investment firm Jefferies predicted for one US airline that, if every passenger flying with it lost roughly 10 pounds (4.5 kilograms), the airline would save more than 100 million litres of fuel per year. Other reports found potential impacts for companies producing medical devices such as knee implants for arthritis or masks for sleep apnoea, more demand for smaller cars, and reconfiguration of land and building use if people walk more in commercial areas.
On a global scale, the ripple effects on economies could be even greater. For instance, studies suggest that young people with obesity perform worse in school1 and that girls are less likely to go on to pursue higher education than their non-obese counterparts are2, even when controlling for factors such as race, income and parents’ socioeconomic level or education. Obesity-related health problems have also been shown to lead workers to take more sick days — which in turn can lead to workplace discrimination.
Costs such as these account for more than 2% of global gross domestic product (GDP), according to a 2022 report on obesity rates in 161 countries3. If the rate of obesity increase was suddenly slowed by 5% relative to current trends, the report found, countries would save more than $429 billion each year between 2020 and 2060. Such analyses do not try to model the costs of drugs or any other intervention that could lead to lower obesity rates.
Unclear consequences
But when it comes to GLP-1 agonists, some of these predictions might be premature, says Ross Hammond, a systems scientist at the Brookings Institution, a think tank in Washington DC, because weight loss with GLP-1 agonists might not directly translate into global cost savings and health improvements. “I’d be very uncomfortable saying the cost estimates [in those studies] are costs that could be saved by everyone being on Ozempic,” he says. “It’s not clear all those consequences would be good.”
For instance, an economic principle known as moral hazard predicts that people tend to behave in risky or unhealthy ways if they face no consequence. It’s not yet clear whether people taking GLP-1 agonists adopt healthier lifestyles if they no longer have to worry about gaining weight, although some evidence suggests that the drugs reduce cravings for high-fat and high-sugar food. The same goes for exercise: research hasn’t yet shown whether people become more or less physically active if they lose weight with GLP-1 agonists.

Cheaper versions of blockbuster obesity drugs are being created in India and China
Hammond points out that exercise and healthy diets have benefits beyond weight maintenance. Reducing caloric intake alone won’t solve issues such as weak bones or muscles, driven by sedentary lifestyles and micronutrient deficiencies. “I’m a little worried that looking for a pharmaceutical solution will not address some of the bigger systems problems that we really face,” he says.
Vos agrees that modelling the health effects of weight loss with GLP-1 agonists isn’t straightforward. It’s unclear, he says, whether a drug that causes people to suddenly lose weight can be compared with an intervention that prevents them from gaining it in the first place. The duration of obesity might affect factors such as arthritis and cardiovascular risk, much as a person’s risk of lung cancer increases with the number of years that they smoked.
Michele Cecchini, a physician and economist at the Organisation for Economic Co-operation and Development (OECD) in Paris, says that there are other unknowns as well. For instance, many people regain weight years after undergoing bariatric surgery. That might turn out to also be the case for people who take weight-loss drugs for decades. “These are things that have a potentially huge impact at the population level,” he says.
Cecchini says that the OECD plans to release a report on GLP-1s’ economic impacts next year. His group has previously looked at the effects of obesity on factors such as education, workforce participation and health-care spending. Even climate change is affected when obesity rates rise. Carbon dioxide levels from food manufacturing rise to meet increased demand — particularly with red-meat production — and vehicles have greater emissions owing to heavier loads4.
The challenge with weight-loss drugs, Cecchini says, is the lack of long-term data. There’s a particular lack of independent studies, because many are funded by the drugs’ manufacturers, he adds.
Disparate outcomes
Some data are more solid than others. Researchers could probably confidently project the drug’s impact in people in their sixties and seventies with high body mass indexes (BMIs), says Zachary Ward, a decision-science researcher at the Harvard T.H. Chan School of Public Health in Boston, Massachusetts. The effects of obesity on conditions such as diabetes, Alzheimer’s disease and certain cancers that affect this age group are well known.
A study of more than 17,000 people that was published last November5 estimated that Wegovy reduced the risk of heart attacks and strokes by 20% in people who had cardiovascular disease and were also overweight. And another research team estimated that 93 million people in the United States could benefit from Wegovy. If these people all took Wegovy, the team calculated, it would prevent 1.5 million cardiac events over ten years6.
But young people taking GLP-1 agonists are unlikely to encounter these diseases for many years, and factors such as lifestyles and environmental exposures can change over decades. “The longer you go, the more assumptions you make,” Ward says.
Some models show that younger people might benefit most from GLP-1 agonists — if they take them for the rest of their lives. Sexton Ward’s group found that, contrary to conventional wisdom, the drugs are less cost-effective for people with very high BMIs7. Rather, they can prevent more diseases in young people with BMIs between 30 and 40 — the lower end of the range for obesity. “That’s the range where it’s effective enough that 20% weight reduction will reduce the risk of comorbidities,” she says.

Obesity drugs aren’t always forever. What happens when you quit?
Their analysis found that, if everyone in the United States had free access to semaglutide, the resulting drop in obesity-associated diseases would save taxpayers $24.5 billion per year, although this would not include the cost of the drugs themselves.
Sexton Ward agrees that predicting population-level health and economic impacts is difficult owing to scarce data. But she highlights one idea: in a report funded by semaglutide-maker Novo Nordisk that she co-authored, her team found that the GLP-1s could produce much greater economic benefits for Black and Hispanic people in the United States than for white populations8.
US Black and Hispanic populations are, on average, more prone to obesity and related conditions than white populations are — largely because of societal inequalities such as lower average incomes and concomitant difficulties in accessing healthy food. Obesity, in turn, exacerbates other health disparities caused by environmental exposures and discrimination in health care.
“Over time, access to these drugs could begin to shrink some of these disparities,” Sexton Ward says. Although obesity isn’t the only cause of health inequality, she says, “I think it could be a step in the right direction.”
The price issue
Right now, however, the drugs are so expensive that only relatively wealthy people can afford them (although in some US states, there is government health-care coverage under a programme for low-income individuals). If influencers and celebrities can easily lose weight, Rasmussen says, the stigma against obesity might increase. That could further harm people on low incomes, who are most likely to face the social, economic and health consequences of obesity. “That gap will increase as long as it remains expensive,” he says.
The high cost is a particular problem for low- and middle-income countries, where obesity rates are growing twice as fast as in high-income nations, says Adeyemi Okunogbe, a health-policy researcher at the non-profit RAND Corporation in Santa Monica, California. Of the estimated 5 million deaths each year caused by obesity-associated conditions, 77% occur in low- and middle-income countries.
Okunogbe says that obesity-related health-care costs put a double burden of disease on these countries, many of which are already fighting higher rates of infectious diseases. Other costs, such as lost wages owing to higher rates of illness and disabilities, are also magnified in lower-income nations. And in countries without national health systems, individuals would need to pay for GLP-1 agonists out of their own pocket. “It’s a long way off to consider this medicine in this context,” Okunogbe says.
Even countries with public health-care systems are struggling to work out how to pay for the drugs. The UK National Health Service currently funds semaglutide for individual weight loss for only two years, even though the drug must be taken for life to sustain its effect. And US law doesn’t require taxpayer-funded insurance programmes to cover weight-loss treatments (although the same drugs can be covered to treat diabetes). The drugs aren’t even close to affordable: an analysis last year9 by Hur’s group found that the cost of semaglutide would need to drop by 85% to make it cost-effective for adolescents. “The way things stand, there’s not going to be much impact on global health outcomes,” Ward says.
Cecchini says that the OECD’s analysis will consider the impact of cheaper generic and compounded GLP-1 agonist drugs, which are beginning to appear on the market in many countries. And some lawmakers, including in the United States, are beginning to push back on the high costs, particularly after an analysis found that Novo Nordisk could sell Ozempic for $5 per month and still make a profit10.
Until more people can afford the drugs, economists and public-health researchers hoping to look at global impacts are stuck with virtual models instead of studies of actual behaviour. “These drugs hold so much promise, but [the world] has to have access to realize it,” Sexton Ward says.
Credit: nature.com
Tirzepatide safe, effective for weight loss, preventing diabetes progression at 3 years
Team Metabolic Health
Tirzepatide demonstrated long-term weight-loss benefits and prevention of diabetes progression in patients with obesity and prediabetes, but only so long as they continued the weight-loss medication, a speaker reported.
The SURMOUNT-1 trial was a randomized, double-blind, placebo-controlled study for which patients with overweight plus one weight-related comorbidity or obesity and without diabetes were randomly assigned to 72 weeks of tirzepatide 5 mg, 10 mg or 15 mg doses (Mounjaro/Zepbound, Eli Lilly) or placebo.

Long-term tirzepatide reduced body weight and diabetes progression and was well tolerated in people with obesity and prediabetes. Image: Adobe Stock
As Healio previously reported, adults with overweight or obesity without diabetes experienced an approximately 20.9% weight loss at 72 weeks with 15 mg tirzepatide.
The top-line results from the SURMOUNT-1 3-year study were previously reported in August 2024.
At ObesityWeek, Leigh Perreault, MD, associate clinical professor of medicine in the division of endocrinology, metabolism and diabetes at the University of Colorado Hospital, moderated a session overviewing the full 3-year efficacy and safety results of the SURMOUNT-1 trial.
The objective of the SURMOUNT-1 3-year trial was to evaluate the impact of tirzepatide on change in body weight and glycemia in participants with prediabetes and whether the drug prevented progression to type 2 diabetes.
“Here in the U.S., according to the CDC, approximately 40 million Americans live with diabetes,” Perreault said during a presentation. “The epidemic in diabetes, whether here in the U.S. or abroad, is clearly being driven in large part by prediabetes.”
Louis J. Aronne, MD, FACP, FTOS, DANOM, the Sanford I. Weill Professor of Metabolic Research at Weill Cornell Medical College, discussed the baseline characteristics of the 1,032 SURMOUNT-1 3-year participants, all of whom were diagnosed with prediabetes upon trial enrollment (mean age, 48 years; 64% women; 73.4% white).
Aronne said all participants also had obesity or overweight with one additional weight-related comorbidity. Diagnosis of prediabetes was defined by the American Diabetes Associations cutoffs and required at least two abnormal tests, according to the presentation.
The trial consisted of a 20-week dose-escalation period. Participants with prediabetes who remained on tirzepatide during the first 72 weeks underwent a further 104-week treatment period on tirzepatide 5 mg, 10 mg or 15 mg for a total treatment period of 176 weeks, after which participants were followed up 17 more weeks off-treatment to assess safety for the totality of trial duration, according to the presentation.
Three-year efficacy of tirzepatide for weight loss
Ania M. Jastreboff, MD, PhD, associate professor of medicine and pediatrics at Yale School of Medicine, director of the Yale Obesity Research Center and co-director of the Yale Center for Weight Management, presented the main efficacy data on tirzepatide for weight loss at 3 years.
Key endpoints included mean percentage change in body weight from randomization to week 176 and time to type 2 diabetes onset at week 176 — during treatment period — and week 193 — during post treatment follow-up.
Among the original SURMOUNT-1 cohort of 2,539 participants, 40.6% had prediabetes at randomization and were eligible to continue into the SURMOUNT-1 3-year trial.
Jastreboff reported that the average 3-year weight reduction with tirzepatide 15 mg was approximately 22.9%, translating to an average weight reduction of 34.6 lb to 54.2 lb, depending on the dose of tirzepatide (based on a baseline weight of 236.8 lb).
During 3 years of follow-up, 12.6% of participants assigned to placebo developed type 2 diabetes compared with 1.2% of participants assigned to tirzepatide, which correlated to an HR for diabetes of 0.06 (P < .001), or a 94% reduction in risk for developing diabetes among patients with prediabetes taking tirzepatide. The reported number of SURMOUNT-1 participants needed to treat to prevent one new case of diabetes was nine, according to the presentation.
During the on-drug treatment period, up to 94.5% of participants assigned to tirzepatide had reversion to normoglycemia compared with 60.4% of those assigned to placebo and lifestyle intervention.
Jastreboff and colleagues estimated that approximately 55.2% of diabetes risk reduction was mediated by the weight-loss effects of tirzepatide.
In addition, participants assigned to tirzepatide 15 mg long term also experienced:
- an average decrease in HbA1c of 0.5% to 0.65%;
- a mean reduction in baseline waist circumference from 45.9 inches to 38 inches;
- an average 8 mm Hg decrease in systolic BP and a 5.9 mm Hg decrease in diastolic BP;
- a more than 14% increase in HDL;
- a more than 32% decrease in triglycerides;
- improvement in all lipid levels; and
- improvement in all eight domains of the Short Form-36v2 quality of life questionnaire.
Upon treatment discontinuation after 176 weeks on tirzepatide, the researchers reported reversals toward baseline in weight, type 2 diabetes risk and blood pressure during the ensuing 17-week off-drug period.
“Treatment with all three doses of tirzepatide demonstrated statistically significant and sustained body weight reduction compared with placebo over more than 3 years,” Jastreboff said during the presentation. “Diabetes prevention with tirzepatide over 3 years resulted in a 94% reduction in progression to diabetes … and nearly 95% reverted from prediabetes to normoglycemia. There was improvement in all cardiometabolic measures. Improvements in all domains of health-related quality of life. Off-drug weight regain was observed and was accompanied by worsening glycemia and increase in progression to type 2 diabetes.”
Three-year safety of tirzepatide for weight loss
Sean Wharton, MD, FRCPC, PharmD, medical director of the Wharton Medical Clinic, adjunct professor at McMaster University in Hamilton, Ontario, and York University in Toronto, academic staff at Women’s College Hospital and clinical staff at Hamilton Health Sciences, presented the main safety results of the 3-year trial.
Wharton reported no significant difference between placebo and any of the tirzepatide doses in mortality rate; however, early discontinuation due to treatment-emergent adverse events was higher among those assigned to tirzepatide compared with placebo.
The most common adverse events reported were gastrointestinal (GI)-related, and those reported were of generally mild to moderate severity. The most common GI-related events included were diarrhea, nausea, constipation and vomiting.
Cholelithiasis was also more frequently observed in the tirzepatide group compared with placebo, a finding consistent with prior studies.
These results were consistent with previously published safety and tolerability of tirzepatide at 72 weeks in SURMOUNT-I and other clinical studies of tirzepatide, according to the presentation.
“The tolerability and safety profile of tirzepatide in this study of 3 years and 9 months duration was generally consistent with incretin-based therapies in people with obesity. The most common adverse events were GI symptoms, generally mind to moderate in severity, occurring primarily during dose escalation. Among adverse events, the most common reasons for discontinuation of the medication were GI adverse events. Gallbladder disease events were reported more frequently in participants from the tirzepatide 10 mg and 15 mg groups compared to the placebo. The finding was mainly due to the increased incidence of cholelithiasis.”
‘Shift focus from weight loss to health gain’
After the presentation of the SURMOUNT-1 3-year safety results, Carel Le Roux, MBChB, MSC, FRCP, FRCPath, PhD, director of the metabolic medicine group at the University College Dublin, summarized the trial as a way to “shift focus from weight loss to health gain.”
“In people with obesity with prediabetes treatment of the disease of obesity, treatment could achieve control of the disease, but not cure. Metabolic complications of obesity got better and stayed better. Functional complications of obesity got better and stayed better. Mental complications of obesity got better but may return close to baseline. Uncertainty remains regarding what happened with appetitive behavior,” Le Roux said. “We can now shift the focus away from weight loss to health gain. When treating patients with prediabetes and obesity we can realize substantial health and functional goals.”
Credit: healio.com
Zealand Pharma to start Phase 2b trial for obesity drug candidate in Q4
Team Metabolic Health
Danish biotechnology company Zealand Pharma (ZELA.CO), said on Thursday it expected to initiate a so-called Phase 2b trial with its obesity treatment candidate petrelintide in the fourth quarter of 2024.
“We look forward to initiating a large, comprehensive Phase 2b trial very soon and are now exploring collaboration opportunities with potential partners,” Zealand Pharma CEO Adam Steensberg said in a statement.
The company said it expected to initiate the trial with petrelintide in people with overweight or obesity without type 2 diabetes in the fourth quarter and to complete enrolment of patients in the first half of 2025.
It also said it also expected to initiate a Phase 2b trial in the first half of 2025 for patients with overweight or obesity and type 2 diabetes.
Credit: Reuters
Eli Lilly gets nod to launch weight-loss drug in Hong Kong – Bloomberg News
Team Metabolic Health
A box of Mounjaro, a tirzepatide injection drug used for treating type 2 diabetes made by Lilly is seen at Rock Canyon Pharmacy in Provo, Utah, U.S. March 29, 2023. REUTERS/George Frey/File Photo
Eli Lilly (LLY.N), opens new tab expects to start selling its weight-loss drug in Hong Kong as early as the end of this year, Bloomberg News reported.
The drugmaker has received approval from the Hong Kong government to sell its tirzepatide injections — branded as Mounjaro — in a device called Kwikpen for both long-term weight management and type 2 diabetes, Eli Lilly told Bloomberg in a statement.
Tirzepatide is the active ingredient in the U.S. firm’s diabetes drug Mounjaro, and weight-loss drug Zepbound.
Eli Lilly did not immediately respond to a Reuters request for comment.
The company had received approval from Chinese regulators for its weight-loss drug tirzepatide in July, further intensifying competition with Danish rival Novo Nordisk (NOVOb.CO), opens new tab in the key Asian market.
Novo Nordisk and Eli Lilly are racing to increase production in a weight-loss market estimated to reach at least $100 billion globally by the end of the decade.
Both companies’ obesity treatments belong to a class of drugs originally developed for diabetes known as GLP-1 agonists.
Credit: Reuters
80% of Strokes Are Preventable and GLP-1 drugs are beneficial for cardiovascular health
Finding to be extremely effective in treating risk factors for stroke, American Stroke Association incorporates the important new class of drug GLP-1 agonists.
Team Metabolic Health
The American Stroke Association has updated its guidelines on stroke risk reduction for the first time since 2014.
In their recommendations, the ASA urges healthcare professionals to carefully screen patients for risk factors such as high blood pressure, elevated cholesterol levels, obesity, and high blood sugar.
The new stroke guidelines highlight unique risk factors for females and transgender women and how social determinants of health impact stroke risk.
The guidelines also emphasize the benefits of GLP-1 drugs for cardiovascular health, and encourage a balanced diet, regular physical activity, maintaining a healthy weight, and getting adequate sleep.
The Centers for Disease Control and Prevention (CDC) estimatesTrusted Source nearly 800,000 people have a stroke every year in the United States. Of these, more than 600,000 will be a first-time stroke.
For the first time in 10 years, the American Stroke Association (ASA) has updated its guidelines for reducing stroke risk. According to an ASA news releaseTrusted Source, up to 80% of strokes may be preventable through diet and lifestyle changes and other modifications.
The 2024 Guideline for the Primary Prevention of StrokeTrusted Source also urges healthcare professionals to carefully screen individuals for stroke risk factors, including: High Blood Pressure, Elevated Cholesterol Levels, High Blood Sugar and Obesity.
Many of the new stroke guidelines coincide with the American Heart Association’s Life’s Essential Trusted Source, which promotes cardiovascular and brain health. The lifestyle factors in the ASA’s new stroke guidelines include a healthy diet, regular exercise, better sleep, and smoking cessation.
“Patients looking to reduce their stroke risk (and cardiovascular disease risk in general) should strive to follow the American Heart Association’s Essential 8 recommendations for healthy living,” said Cheng-Han Chen, MD, an interventional cardiologist and medical director of the Structural Heart Program at MemorialCare Saddleback Medical Center in California. Chen wasn’t involved in the new guidelines.
“We recommend that people eat a healthy balanced diet, engage in regular physical activity, get an adequate amount of sleep, maintain a healthy weight, avoid tobacco and alcohol, reduce stress, and control their blood pressure and cholesterol numbers,” Chen told Healthline.
Overall, the guidelines provide a clear pathway for medical professionals and their patients. The guidelines also call for more public awareness and education about how people can lower their risk of stroke.

New guidelines for stroke prevention from the American Stroke Association emphasize the benefits of GLP-1 drugs, a balanced diet, regular physical activity, and maintaining a healthy weight. PIKSEL/Getty Images
Women face unique stroke risk factors
The updated stroke guidelines recommend that medical professionals screen for conditions that can increase the risk of stroke in females.
Stroke risk factors affecting females include the use of oral hormonal contraceptives, developing high blood pressure during pregnancy, and pregnancy complications, such as: Premature birth, Endometriosis, Premature ovarian failure and Early-Onset Menopause.
The guidelines also note that transgender women and gender-diverse individuals taking estrogen may also be at an increased risk of stroke.
Social determinants of health and stroke risk
The new stroke recommendations emphasizeTrusted Source “social drivers of health.” These non-medical factors include inequities in healthcare, education, economic stability, and structural racism.
The ASA adds that neighborhood factors such as a lack of walkability and access to healthy food also play a role. The guidelines urge medical professionals to advocate for patients in these circumstances.
“Some populations have an elevated risk of stroke, whether it be due to genetics, lifestyle, biological factors and/or social determinants of health, and in some cases, people do not receive appropriate screening to identify their risk,” said Cheryl Bushnell, MD, a professor and vice chair of research in the Department of Neurology at Wake Forest University School of Medicine in North Carolina, in a news releaseTrusted Source. Bushnell is chair of the group that wrote the new guidelines.
“This guideline is important because new discoveries have been made since the last update 10 years ago,” she continued. “Understanding which people are at increased risk of a first stroke and providing support to preserve heart and brain health can help prevent a first stroke.”
José Morales, MD, a vascular neurologist and neurointerventional surgeon at Providence Saint John’s Health Center in California Morales echoed the importance of primary stroke prevention and said the guidelines were long overdue. Morales wasn’t involved in developing the new guidelines.
“The focus on equitable access to healthcare, social determinants of health, and gender-specific recommendations are important additions to guiding clinical practice in an evidence-driven matter,” he told Healthline.
GLP-1 drugs may reduce stroke risk
The ASA’s guidelines include a new recommendation for healthcare professionals to consider prescribing GLP-1 weight loss drugs, such as Ozempic, Mounjaro, Zepbound, and Wegovy.
The medications, which have been approved by federal regulators as treatments for obesity or type 2 diabetes, have shown promise in clinical trials in improving cardiovascular health and reducing the risk of stroke.
Chen said the inclusion of newer topics such as weight-loss drugs into the new guidelines is an important step.
“These updated guidelines also incorporate the important new class of drug (GLP-1 agonists) that we are finding to be extremely effective in treating those same risk factors for stroke,” Chen noted.
Healthy diet for stroke prevention
One of the most important things you can do to prevent a stroke is to maintain a healthy, balanced diet.
The new guidelines urge people to stick closely to a Mediterranean diet. This diet focuses on the eating habits of people who live along the Mediterranean Sea.
That plan encourages people to consume more fruits and vegetables as well as legumes, nuts, seeds, and heart-healthy fats. It discourages the consumption of processed foods, added sugars, refined grains. It also recommends limiting the consumption of alcohol.
“Risk factors for stroke vary by age, gender, genetics, comorbidities, and even medications. Therefore, I think the suggestion of screening for not only the basic risk factors but also secondary factors is essential to prescribe the right lifestyle interventions,” said Kristin Kirkpatrick, MS RDN, a dietitian at the Cleveland Clinic Department of Wellness & Preventive Medicine in Ohio. Kirkpatrick wasn’t involved in the new guidelines.
“For my patients, I will often also recommend a Mediterranean pattern, but then work with the patient to assess tweaks to that pattern,” she told Healthline.
“For example, with someone who has insulin resistance or type 2 diabetes, I may focus on a more moderate carbohydrate version of the Med diet. For patients with high blood pressure, I may counsel more on a low sodium perspective. The most important component, however, may be to focus as a whole on a more plant-forward approach that limits ultra-processed foods, sugar, refined grains, and excess amounts of alcohol,” Kirkpatrick continued.
“I also recommend nutrigenomics testing for my patients as well to be able to get a truly personalized approach to prevention based on genetics,” she said.
Exercise and stroke prevention
The guidelines recommend that healthcare professionals screen patients for sedentary behavior and counsel them to engage in regular exercise.
As noted by the ASA’s news releaseTrusted Source, physical activity is “essential for stroke risk reduction and overall heart health.”
The CDC recommendsTrusted Source that adults get at least 150 minutes a week of moderate-intensity aerobic activity or 75 minutes a week of vigorous aerobic activity. That exercise should be spread throughout the week.
Regular exercise can help improve metrics such as blood pressure, cholesterol levels, inflammatory markers, insulin resistance, and overall weight.
“A combination of both aerobic and anaerobic is essential to focus on both muscle retention (and growth) as well as cardiovascular health. Flexibility is also an important factor,” Kirkpatrick said.
Credit: healthline.com
Can weight loss drug semaglutide slow down ageing? Here’s looking at its multiple benefits
Team Metabolic Health
Given their effects on various organs, they could potentially help you remain functional
“New diabetes drugs may delay ageing” made headlines across the media. The reference was to a study using glucagon-like peptide receptor agonist (GLP1RA), which mimics the hormone incretin to regulate appetite and blood glucose levels. Semaglutide, popularly known as Ozempic, belongs to this class of drugs and is known for controlling blood sugar, suppressing hunger and reducing weight. Are they truly anti-ageing, too?
What is ageing?
Ageing is a highly complex biological process. Our DNA is constantly being broken down and repaired. An imbalance in this process, leading to poor repair of DNA, is one of the hallmarks of ageing. Telomere shortening, deranged protein metabolism and altered sensing of nutrients are other features. These changes lead to cellular senescence (cells lose their power to grow and multiply), reduced tissue regeneration and reduced communication between cells.

Some of the “anti-ageing” benefits seen with GLP1RAs may simply be the result of their weight-loss inducing properties. (File image)
What’s the link between weight loss drugs and ageing?
GLP1RAs have been shown to protect against oxidative stress, cellular senescence and chronic inflammation, which are widely accepted as the major risk factors of ageing. This is characterised by memory loss, cognitive impairment and coordination deficits. Recent studies suggest that GLP1RAs could have beneficial neuroprotective effects. A large study from Sweden showed that in people with Type 2 diabetes, the use of GLP-1RA was associated with 23-30 per cent lower risk of dementia, compared with other anti-diabetic agents.
There are several common pathological processes in Alzheimer’s Disease, a brain disorder that causes a gradual decline in memory and thinking skills, and Type 2 diabetes. These include impaired glucose metabolism, insulin resistance, increased oxidative stress and increased inflammation. Alzheimer’s Disease has been referred to as diabetes of the brain.
What’s their efficacy in managing diseases?
GLP1RAs have shown great promise in treating Alzheimer’s and have also shown some efficacy in treating Parkinson’s disease. Definitive, larger studies are ongoing on this subject. Some of the “anti-ageing” benefits seen with GLP1RAs may simply be the result of their weight-loss inducing properties. Obesity promotes the development of Type 2 diabetes, heart disease and early death. Reduction in body weight can prevent, delay or reduce the development of these conditions with advancing age.
GLP-1RAs, including semaglutide, have been shown to reduce the risk of cardiac complications in people with Type 2 diabetes. The SELECT trial tracked more than 17,600 people, aged 45 or older, as they were given either 2.4 mg of semaglutide or a placebo for more than three years. Participants were obese or overweight and had cardiovascular disease but were not diabetic. Men and women who took the drug died at a lower rate from all causes, including cardiovascular issues and Covid-19. People using the weight-loss drug were less likely to die from Covid (2.6 per cent vs 3.1 per cent). Heart failure, too, was lower in the semaglutide group.
Results of the FLOW trial published earlier this year showed that semaglutide slowed the progression of kidney disease in people with diabetes by 24 per cent. Studies have further shown a reduction in fatty liver disease with GLP-1RAs. Reduction in liver fat could reduce chances of chronic liver disease and its serious, at times fatal consequences.
One concern with the use of GLP1RAs is the muscle loss that accompanies profound weight loss — such muscle loss can potentially lead to frailty, falls and fractures, and prove to be a negative impact of this class of drugs in the elderly.
Credit: The Indian Express
The Novo Nordisk scientist behind Ozempic, Wegovy weight loss research
Team Metabolic Health
Credit: You Tube Channel 60 Minutes
AstraZeneca says experimental obesity pill safe in early-stage trial
Team Metabolic Health
AstraZeneca (AZN.L), opens new tab said on Monday its experimental weight-loss pill, licensed a year ago from China’s Eccogene for up to $2 billion, was safe and tolerable in an early-stage trial, with side effects consistent with the GLP-1 drug class.
Sharon Barr, executive vice president of biopharmaceuticals R&D, said AstraZeneca had progressed the drug into Phase II clinical trials based on the data from Phase I, which assessed its safety and tolerability in 72 participants who were either healthy, non-obese volunteers or people with type 2 diabetes.

The logo for AstraZeneca is seen outside its North America headquarters in Wilmington, Delaware, U.S., March 22, 2021. REUTERS/Rachel Wisniewski/File Photo
AstraZeneca’s shares rose as much as 2.9% after the data release, before paring gains. When AstraZeneca announced it had licensed the once-daily pill, called AZD5004, it said it believed the treatment could cause fewer side effects than injectable treatments such Eli Lilly’s (LLY.N), opens new tab Zepbound and Novo Nordisk’s (NOVOb.CO), opens new tab Wegovy.
One of the Phase II trials of the drug, will focus on reduction in body weight in obese and overweight participants and is expected to be completed by the end of 2025, Barr said.
“We saw a dose-dependent increase in nausea and vomiting consistent with molecules in this class,” Barr told a briefing, referring to GLP-1 receptor agonists, which slow digestion and reduce hunger. Zepbound and Wegovy are both from this class.
There were no serious adverse events reported, she said.
CEO Pascal Soriot acknowledged after the Eccogene deal that AstraZeneca was “a few years behind” Novo Nordisk and Eli Lilly who were first to market with highly effective obesity drugs.
The drugmaker is now bullish for its obesity business.
Its weight-loss pill compares favourably to others in clinical development by rival companies because it is a small molecule that can be combined with other small molecule drugs, said Barr, which is important because more than 60% of obese and overweight people have one or more other medical conditions.
The pill can be taken with or without food, Barr added, citing data released from an early-stage trial of 14 patients.
AstraZeneca also released data from Phase I trials of its other leading experimental obesity drugs, which are injected.
One, AZD6234, targets a pancreas hormone called amylin that affects hunger. Another, called AZD9550, targets the gut hormone GLP-1 and a second obesity-related hormone called glucagon.
Trials showed both were safe and tolerable, AstraZeneca said, adding it had begun Phase II trials in overweight and obese patients for AZD6234 and expects to complete in 2026. It will start them for AZD9550 in the next six months.
Meanwhile, Viking Therapeutics (VKTX.O), opens new tab on Monday published results from an early-stage trial of its oral obesity treatment that analysts said compared well to rivals in development.
Credit: Reuters
