Children with Obesity Have Increased Risk of Atopic Dermatitis
Team Metabolic Health
A new study revealed children who go from normal weight to overweight have a greater risk of atopic dermatitis than obese children who maintain their weight.
A new study found children with obesity have an increased risk of pediatric immune-mediated skin diseases, particularly atopic dermatitis. Conversely, weight loss can lower the risk.
“This is the noteworthy longitudinal investigation that has revealed the association between BMI changes and subsequent occurrences of 3 main pediatric [immune-mediated skin diseases]—[alopecia areata, atopic dermatitis, and psoriasis]—as well as the direct influence of BMI on [alopecia areata],” wrote co-lead investigators Hyunsun Park, MD, PhD, Seong Rae Kim, MD, and Seong-Joon Koh, MD, PhD, from Seoul National University College of Medicine.
In recent years, immune-mediated skin diseases, such as alopecia areata, atopic dermatitis, and psoriasis have become a public health concern among children. These conditions harm the quality of life regarding emotional, physical, social, and functional well-being.
Obesity rates have also surged in the previous years. Research has suggested excessive adipose tissues may trigger proinflammatory mechanisms that lead to the development of skin diseases, however, this topic still needed further investigations as this was speculation. In this study, investigators wanted to understand better how childhood obesity or weight gain interacted with the development of pediatric immune-mediated skin diseases.
“Our research team is very interested in the skin-gut access,” said co-lead investigator Hyunsun Park, MD, PhD, from the department of dermatology at Seoul National University College of Medicine in Seoul, Republic of Korea. “We think that various factors, including diet, obesity, or other lifestyles can affect gut environment and contribute to the development of IMSDs. We are trying to find the puzzle pieces to demonstrate how they are connected, and our current research is a step towards that understanding.”
Thus, investigators, also co-led by Seong Rae Kim, MD, and Seong-Joon Koh, MD, PhD, both from the Seoul National University College of Medicine, conducted a study to assess the associations between body mass index (BMI) or BMI changes and the development of 3 immune-mediated skin diseases: alopecia areata, atopic dermatitis, and psoriasis.
The team analyzed a longitudinal cohort of 2,161,900 Korean children from 2009 to 2020. The sample included 10,524,561 person-years in the alopecia areata cohort, 6,910,806 person-years in the atopic dermatitis, and 10,530,018 person-years in the psoriasis cohort. Overall, the cases of immune-mediated skin diseases were 4,878 for alopecia areata (51.1% male), 41,386 for atopic dermatitis (50.4%); and 2,191 for psoriasis (51.1).
The findings demonstrated children with obesity had a greater risk of pediatric immune-mediated skin diseases than those with a normal weight (P < .01). Children who had an increase in BMI with a weight gain from normal to overweight had a greater risk of atopic dermatitis (adjusted hazard ratio [aHR], 1.15; 95% confidence interval [CI], 1.11 – 1.20). Reversely, a decrease in BMI—and weight loss from overweight to normal weight, had a lower atopic dermatitis risk than children who were overweight and maintained their weight (aHR, 0.87; 95% CI, 0.81 – 0.94; P < .001).
“Our findings support the importance of promoting weight maintenance among children who are already within the normal weight range because it may help reduce the risk of developing atopic dermatitis,” Koh said in the press release. “In addition, prevention of excessive weight gain and purposeful weight loss, including adopting healthy diet strategies in children with obesity to prevent atopic dermatitis, particularly before school age, should be promoted. Implementing purposeful interventions, including nutritional strategies, to decrease body weight may aid in reducing the risk of developing [immune-mediated skin diseases] in children.”
Credit: https://www.hcplive.com/
New method accelerates drug discovery from years to months
Team Metabolic Health
Researchers from the University of Cincinnati College of Medicine and Cincinnati Children’s Hospital have found a new method to increase both speed and success rates in drug discovery.
The study, published Aug. 30 in the journal Science Advances, offers renewed promise when it comes to discovering new drugs.
“The hope is we can speed up the timeline of drug discovery from years to months,” said Alex Thorman, PhD, co-first author and a postdoctoral fellow in the Department of Environmental and Public Health Sciences in the College of Medicine.
Researchers combined two approaches for screening potential new drugs. First, they used a database from the Library of Integrated Network-based Cellular Signatures (LINCS) to screen tens of thousands of small molecules with potential therapeutic effects simultaneously. Then they combined the search with targeted docking simulations used to model the interaction between small molecules and their protein targets to find compounds of interest. That sped up the timing of the work from months to minutes -; taking weeks of work required for initial screening down to an afternoon.
Thorman said this faster screening method for compounds that could become drugs accelerates the drug research process. But it’s not only speed that is crucial.
He added that this newer approach is more efficient at identifying potentially effective compounds.
It can also create more targeted treatment options in precision medicine, an innovative approach to tailoring disease prevention and treatment that takes into account differences in people’s genes, environments and lifestyles.
“An accelerated drug discovery process also could be a game changer in the ability to respond to public health crises, such as the COVID-19 pandemic,” said Thorman. “The timeline for developing effective drugs could be expedited.”
The other co-first authors were Jim Reigle, PhD, a postdoctoral fellow at Cincinnati Children’s Hospital, and Somchai Chutipongtanate, PhD, an associate professor in the Department of Environmental and Public Health Sciences in the College of Medicine.
The corresponding authors of the study were Jarek Meller, PhD, a professor of biostatistics, health informatics and data sciences in the College of Medicine, and Andrew Herr, PhD, a professor of immunobiology in the Department of Pediatrics in the College of Medicine.
Other co-investigators included Mario Medvedovic, PhD, professor and director of the Center for Biostatistics and Bioinformatics Services in the College of Medicine, and David Hildeman, PhD, professor of immunobiology in the College of Medicine. Both Herr and Hildeman have faculty research labs at Cincinnati Children’s Hospital.
This research was funded in part by grants from the National Institutes of Health, a Department of Veterans Affairs merit award, a UC Cancer Center Pilot Project Award and a Cincinnati Children’s Hospital Innovation Fund award.
Those involved in the research are also co-inventors on three U.S. patents that are related to their work and have been filed by Cincinnati Children’s Hospital.
Credit: University of Cincinnati
Pigs to the rescue: Revolutionizing kidney transplants
Team Metabolic Health
The numbers are staggering; right now, about 100,000 people are on a waitlist for a kidney transplant but there aren’t enough donors. While they haven’t perfected it, researchers are working to see if some animals might be able to fill the gap.

Pigs to the rescue: Revolutionizing kidney transplants© IVANHOE
When it comes to saving lives for people with kidney disease, the math just does not add up.
“When you think about 37 million Americans with chronic kidney disease and at any given time, [600,000] to 800,000 have actual kidney failure. And we only waitlist [80,000] to 100,000 and we only transplant 25,000,” said Jayme E. Locke, MD, MPH, Arnold G. Diethelm MD Endowed Chair in Transplantation Surgery at the University of Alabama at Birmingham, Heersink School of Medicine.
Researchers at the University of Alabama at Birmingham are some of the first to fill the need with xenotransplantation.
“The goal, of course, is to sort of really basically eliminate that gap between supply and demand. And in our case, we’re hoping to do that with pig organs. Well interestingly, pigs actually have kidney function that’s quite similar to human,” Locke said.
Researchers use a CRISPR, which is a genetic editing tool, to edit out genes that would cause a human body to reject the organ.
“It kind of tricks the human immune system into thinking it’s something from a human,” Locke adds.
And because pigs can live 30 years, researchers believe the organ will last that long for humans, and they hope to use more organs from pigs.
“And so, I think, certainly, those of us in xenotransplantation see a future in which one pig can donate a heart, a pair of lungs, a liver, a kidney, maybe even a pancreas,” Locke said.
Several pig organ transplant recipients have died in recent months following their operation. It’s important to remember that xenotransplantation is still very much in the experimental stages, but researchers are committed to perfecting this alternative source of organs when human organs are not available. The next step will be a clinical trial to seek FDA approval.
Credit: msn.com
Phase 2 Data Supports the Effectiveness, Safety of NBI-1117568 for Schizophrenia
Team Metabolic Health
A phase 2 trial met its primary endpoint, showing NBI-1117568, the first M4 selective agonist, is effective and safe at treating adult schizophrenia. Neurocrine Biosciences recently announced their phase 2 trial met its primary endpoint, demonstrating the effectiveness of NBI-1117568 for schizophrenia. The once-daily 20 mg dose of NBI-1117568 provided a statistically significant 7.5-point improvement in the Positive and Negative Syndrome Scale (PANSS) Total Score compared to the placebo at week 6 (P = .011; effect size of 0.61) with an 18.2-point PANSS Total Score Improvement from Baseline.

Eiry W. Roberts, MD Credit: LinkedIn
“This Phase 2 dose-finding study delivered on our goal of identifying a once-daily, well tolerated dosing regimen with a compelling and competitive benefit-risk profile,” said Eiry W. Roberts, MD, Chief Medical Officer at Neurocrine Biosciences, in a statement. “We recognize the significant need for new and innovative medicines to treat schizophrenia and look forward to advancing NBI-‘568, the first M4 selective agonist, into Phase 3 development early next year.”
According to the World Health Organization, approximately 20 million people worldwide experience schizophrenia. Traditional treatment options for schizophrenia often rely on antipsychotic medications which may lead to short- and long-term health impacts. Thus, investigators wanted to study a novel treatment option for schizophrenia.
Investigators conducted a phase 2, multicenter, randomized, double-blind, placebo-controlled, multi-arm, multi-stage inpatient dose-finding study to assess the efficacy, safety, tolerability, and pharmacokinetics of NBI-1117568 compared with placebo in adult patients with schizophrenia. The study included 210 participants with schizophrenia who experienced an acute exacerbation or relapse of symptoms.
Other than meeting the primary endpoint, the once-daily 20 mg dose of NBI-1117568 also provided significant improvement for secondary endpoints, including the Clinical Global Impression of Severity (CGI-S) scale, Marder Factor Score – Positive Symptom Change, and Marder Factor Score – Negative Symptom Change.
The trial showed NBI-1117568 was safe and well-tolerated at all doses. Investigators observed similar treatment discontinuation rates due to adverse events between participants on NBI-1117568 and placebo. The most common adverse events observed included somnolence, dizziness, and headache.
Less common adverse events included nausea and constipation, cardiovascular-related events, and extrapyramidal symptoms. Gastrointestinal adverse events were similar in both arms, and cardiovascular-related events did not seem to have clinical relevance at any dose. NBI-1117568 was not associated with greater weight gain when compared to placebo.
“NBI-1117568 demonstrated a clinically meaningful and statistically significant reduction in PANSS scores and was well tolerated, importantly with minimal GI effects and no weight gain relative to placebo,” said Maurizio Fava, Psychiatrist-in-Chief at Massachusetts General Hospital of Harvard University, in the press release. “As a selective M4 orthosteric agonist, the potential of NBI-1117568 as an option that could reduce symptoms of schizophrenia with fewer side effects would be a welcome alternative to current treatments for patients and caregivers.”
Credit: hcplive.com
Pfizer, Flagship Pioneering add Quotient Therapeutics to long-term pact
Team Metabolic Health
Pfizer will work with the Flagship-funded Quotient Therapeutics to conduct research that will inform drug development for cardiovascular and renal diseases.
More than a year after Pfizer and venture capital company Flagship Pioneering struck a partnership investing in 10 life sciences programs, the two unveiled a collaboration with Quotient Therapeutics.
The Flagship-funded Quotient, which specializes in somatic genetics — or the study of mutations in somatic cells — will work with Pfizer to conduct research that can inform drug development for cardiovascular and renal diseases. Quotient was founded by Flagship in 2022. Financial terms of the deal were not disclosed.
Flagship’s drug discovery and development unit Pioneering Medicines will be responsible for spearheading the partnership with Pfizer, the company said in a press release. Quotient CEO Jacob Rubens the company’s Somatic Genomics platform as having the potential to investigate the “extensive genetic diversity” among 30 trillion cells in the body. He added that Pfizer’s involvement in these dual programs will allow for the research to scale.

Source: Getty Images
“This offers an incredibly rich and uncharted territory for drug discovery,” Rubens said in a statement. “These two research programs will deploy our unique platform capabilities to identify novel links between genes and cardiovascular or renal disease, which are leading causes of mortality and morbidity around the world.”
The collaboration is the latest update in the Pfizer and Flagship alliance, which was struck in July 2023 to develop a 10-program pipeline of innovative medicines to address unmet needs.
The pact, which involved both companies investing a total of $100 million upfront initially, allows Pfizer to choose from Flagship’s large ecosystem of 40 biotech companies to explore potential therapies.
In June, Pfizer and Flagship announced their first choice — a partnership with ProFound Therapeutics to investigate new weight loss drugs.
That collaboration entails ProFound using its proprietary ProFoundry Platform in combination with Pioneering Medicine’s drug development expertise to explore novel proteins as potential obesity therapies.
At the time, Charlotte Allerton, head of discovery and early development at Pfizer, noted that the partnership was designed to push the “boundaries of science to potentially unlock new protein therapeutics for obesity.”
The announcement also comes a day after Pfizer unveiled its direct-to-consumer (DTC) platform, PfizerForAll, following in the footsteps of Eli Lilly and Company in launching telehealth services.
The goal is for consumers to access a one-stop-shop directly from the big pharma company to book in-person or telehealth appointments, receive home delivery for prescription drugs or schedule vaccinations.
Pfizer launched the platform a little less than a year after Lilly debuted its own DTC platform, LillyDirect.
Credit: mmm-online.com
Full-body scans to look for hidden disease are a bad idea – here’s why
Team Metabolic Health
For a not insubstantial fee, many private clinics in the UK offer full-body MRI scans to detect disease and abnormalities at the earliest stage. Kim Kardashian and Kate Hudson are fans.
So how do these scans work and are they “life saving”, as Kardashian suggests?
MRI produces 3D detailed anatomical images of almost every internal structure in the human body, including the organs, bones, muscles and blood vessels. MRI scanners create images of the body using a large magnet and radio waves. Unlike X-rays or CT, it doesn’t rely on ionising radiation, which can very slightly increase the risk of cancer.
Prices of full body scans in the UK range from £1,000 to £3,000. While they give a good basic overview of your anatomy, they don’t optimise the view of each organ or system. Any unusual findings would need to be further investigated by specialised MRI scans for things such as the heart and prostate. This is because the machine needs to be adjusted to different parameters to optimise the imaging of different organs and systems.
It’s important to note that MRI scans cannot detect most preventable diseases. Heart disease, high blood pressure, high cholesterol and diabetes are responsible for most deaths. MRI scans cannot detect these conditions. They may show some structural changes, but other symptoms will probably have emerged first.
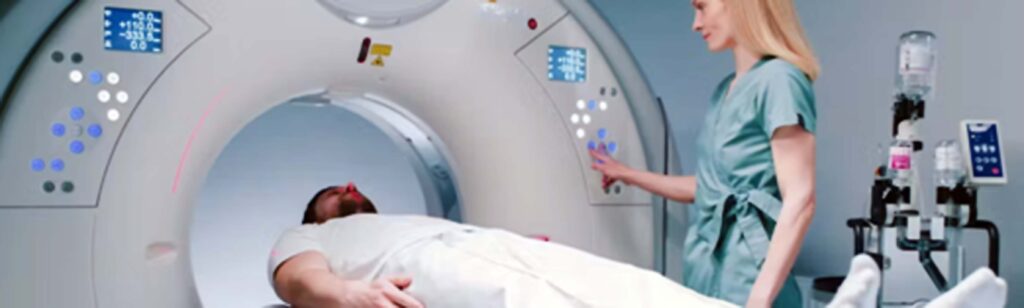
Full-body MRI scans may find abnormalities, often referred to as “incidentalomas” because they are just incidental findings, on scans being undertaken for other symptoms.
Studies looking at over 16,000 brain MRI scans of volunteers have shown that less than 4% of them contained abnormalities that required further investigation.
The kind of things found on brain scans include pituitary abnormalities, such as non-cancerous growths. Disrupted blood flow to the brain (without symptoms) has been detected in 7%, aneurysms in 1.8% and benign tumours in 1.6% of the population.
Small clusters of abnormal blood vessels, known as cavernomas, rarely cause symptoms and are seen in about one in 600 people on MRI scans. Those that do cause symptoms are seen in about one in 400,000 people.
Another area of the body that often throws up incidental findings is the spine. There is increasing use of MRI in back pain. This can lead to the discovery of incidentalomas such as harmless bone growths and haemangiomas, small growths of blood vessels on or in the spine.
Haemangiomas are seen in over 40% of people who receive MRI scans, 99% of which cause no symptoms and don’t need treatment. The 1% who have symptoms, such as pain, numbness or weakness, require surgery to prevent progression towards paralysis.
Tarlov cysts, an outpouching around nerves leaving the spinal cord in the sacral region (near the bottom of the spine) are seen in about 4% of the population. For most, there are no symptoms. But those who have symptoms, such as pain and numbness, may need surgery to prevent bone decay or damage to the nerve supply to the legs, pelvic structures and genitals.

Kim Kardashian wrote about her full-body scan on Instagram. Imago/Alamy Stock Photo
What about serious findings?
A meta-analysis (a study that combines data from many studies) suggests that serious problems that are picked up by chance on MRI scans are 1.4% for the brain, 1.3% for the chest and 1.9% for the abdomen.
There is also a risk that these scans give a false positive – things that look like disease but aren’t. Breasts (up to 97 false positives per 1,000 scans) and prostate (29 per 100 scans) are particularly susceptible to false positive results.
These scans may give some valuable information about what is going on beneath your skin. However, for many people, the reports and terminology used in them are hard to fathom. There is lots of evidence that the average person struggles to understand common medical phrases and often assign meaning opposite to what the doctor intended – for a single diagnosis. Reports resulting from these scans are about the whole body and will contain significantly higher levels of medical information than a single diagnosis – even when everything is normal.
Where people pay for these scans without the wherewithal to cover any follow-up medical investigation, it will be already overstretched healthcare systems that have to pick up the fallout and ensure that false positives and benign growths are confirmed as such.
These scans offer an opportunity to see what’s going on internally – for those with enough money to afford them – but the likelihood of finding something that needs urgent medical treatment is low. If I had the money to spend on one of these, I’d spend it on a holiday, which would be far better for my health.
Credit: The Conversation
A New Drug Could Change The HIV Prevention Landscape, But Only With A Fair Price Tag
Team Metabolic Health
In June, Gilead Sciences announced impressive early results from its PURPOSE 1 trial, which showed 100 percent efficacy of lenacapavir in preventing new HIV infections among cisgender women in Sub-Saharan Africa. Lenacapavir is being investigated as an injectable antiretroviral medication that can be taken once every six months as preexposure prophylaxis (PrEP) to prevent HIV. This is a marked shift from current PrEP options, which are only available as a once-daily pill or a bimonthly injection.
Once again, the HIV community finds itself at a scientific breakthrough moment. And once again, it remains an open question as to whether it will also prove to be a breakthrough moment for equitable access.
We have been down this road before. It was not so long ago that the very same drug manufacturer released similarly jaw dropping clinical trial results first for Truvada and then Descovy, the first two medications approved for the prevention of HIV; it led to widespread speculation of the end of HIV as an epidemic. And yet, more than a decade after the first approval for PrEP in the United States, we still see significant disparities in getting PrEP to communities of color and cisgender women in the US.
Health care advocates are already concerned that past will once again be prologue when it comes to this new PrEP modality, and there is pressure on the company to make commitments that could change all too predictable outcomes on disparate outcomes.
The $40,000 Question
The big question of course will be the price set for lenacapavir’s PrEP indication in the US. We already have some insight here. In May 2019, Gilead received Food and Drug Administration (FDA) approval for a limited HIV treatment indication for lenacapavir for individuals for whom other HIV treatment regimens have not worked. The drug is sold under the brand name Sunlenca, which Gilead launched with a list price of $42,250 per year. To put the Sunlenca price tag in context, the list price of Gilead’s leading brand-name oral medication for PrEP, Descovy, comes in at about $26,000 per year. The bimonthly injectable PrEP option made by Gilead’s competitor, ViiV Healthcare, comes in at a comparable $23,000 per year.
There is no reason to think that as the indication of lenacapavir expands to PrEP, Gilead will back down from its initial high price. And again, we don’t need to guess here, we need only look at past actions. The pricing strategy that Gilead used to set the whopping $1,000-per-pill list price for its curative treatment for hepatitis C was fully explicated in a congressional oversight report, which found that Gilead’s goal was “maximizing revenue, regardless of the human consequences.”
To be clear, the list price is and has always been a bit of a fiction. With the availability of rebates and discounts and low-income assistance programs, few payers or even consumers will ever pay close to that amount. But it does matter. The list price sets the ceiling from which the complex set of drug distribution players in the US negotiate a lower price. Start high and even with discounts, we’ll end high. For a drug with such public health significance as PrEP, the high list price also hamstrings the ability of state and local public health departments that perennially struggle with anemic budgets—some of whom qualify for discounts and some of whom do not—to purchase the drug and get it out to uninsured, low-income, and vulnerable populations.
Other innovative purchasing strategies, such as a subscription model in which safety-net programs pay manufacturers a set negotiated amount for unlimited access to a drug, may be another consideration for PrEP. Subscription models have gotten some traction as a way for state Medicaid programs to cover costly hepatitis C curative treatments, but much more work has to be done to assess how a subscription model might be applied to PrEP, where the volume is lower and where there are multiple effective regimens, including low-cost generics, on the market.
Finally, the high list price locks us into fragmented delivery from the very beginning. Low-income individuals without insurance will be dependent on manufacturer patient assistance to help them access lenacapavir. Those programs will only provide access to the drug and not any other services people need to actually take the drug, including the recommended HIV, sexually transmitted disease, and other testing recommended to safely take PrEP. So instead of a public health program that is able to provide integrated access to PrEP services—including the medication—consumers will have to navigate a web of disparate programs to patch together all the services they need.
As we’ve seen with numerous other new-to-market drugs, a high list price for lenacapavir for PrEP will profoundly limit access for millions of uninsured Americans—a group that is disproportionately Black, Brown, and transgender and gender nonconforming. As noted above, the price point forecloses a public health response to PrEP, forcing individuals to jump through complex hoops to cobble together access. The decision on price made now could set the stage for more than a decade of inequitable access; Gilead has indicated in its most recent 10-K filing (page 7) that it expects lenacapavir to remain on patent and face no significant competition until 2037.
But Wait, Does Research And Development Justify A High Price?
Developing a new drug is an expensive endeavor. And indeed, high price tags are often justified by pointing to the immense costs of research and development. To be fair, Gilead undoubtedly invested in its PURPOSE trials, which have been far more comprehensive than the trials the company oversaw to secure a PrEP indication for Descovy, for which Gilead primarily conducted research among White gay and bisexual men and completely excluded cisgender women and transgender men.
But the cries that high prices are necessary for innovation seem somewhat disingenuous when they are rarely backed by transparency around the funding mix that goes into research and development of any one drug. And such claims deserve scrutiny when researchers have recently shown that lenacapavir could be made for less than $40 per person each year.
Past, again, may be prologue here. According to a Senate Finance Committee report on the development and marketing of Sovaldi, the first curative therapy for hepatitis C, “Gilead failed to provide costs attributable solely to the development of” its blockbuster hepatitis C cure despite repeated requests from Congress. The company from which Gilead acquired sofosbuvir estimated that the final Phase 3 studies would cost only $125.6 million, nowhere near an amount that would justify a $1,000-per-pill price tag. Following the December 2013 FDA approval, Gilead recouped its research costs and then some with $10.3 billion in 2014 sales of stand-alone sofosbuvir along with $2.1 billion in 2014 sales for Harvoni, a single tablet regimen of sofosbuvir combined with another direct-acting antiviral.
Recent research has also unearthed a very different investment story supporting the research and development for Gilead’s first approved PrEP medication, Truvada, finding that the federal government invested $143 million in the research leading to Truvada’s approval, far more than initially estimated and undermining Gilead’s justifications for steep price increases for Truvada following its July 2012 FDA approval as PrEP.
To be fair, developing breakthrough medications requires investment, and manufacturers should have incentives to embark on the long and arduous process of drug development that more often than not leads to a lot of failures before success. However, we should interrogate assertions that any policy aimed at identifying a fair price will imperil research and development incentives. Researchers have directly contradicted this assertion, modeling the impact of federal drug pricing reform and finding that the impact on new drugs coming to market is minimal.
What Can We Do Differently?
There is always hope we will learn from our past mistakes and do things differently the next time around. Advocates and academics alike have been calling for a federally funded national PrEP program to ensure widespread access to PrEP, particularly for un- and underinsured individuals. President Joe Biden included the program in his last three budget requests to Congress; it would provide the infrastructure necessary for the government to work meaningfully with Gilead to come up with an access strategy for lenacapavir at a price point that would work for a public health response. Should that come to pass, the US could really see the potential of this innovation for communities other than cisgender White men.
But even in a scenario in which the government steps in to work with Gilead to solve the access problem for uninsured populations, this pricing will create the same challenges for insured populations that have always existed in the US. Insurers will be absolutely disincentivized to encourage new PrEP users to access lenacapavir, particularly with affordable daily oral generic options at less than $20 per month. And with a price point for lenacapavir $10,000 to $15,000 above Descovy per year, we can anticipate payers’ strong resistance to even transitioning existing PrEP users over to lenacapavir. Even with a US Preventive Services Task Force Grade A for PrEP, which because of an Affordable Care Act (ACA) provision requires most private insurance plans to cover PrEP without cost sharing, there is no mandate for private insurance plans to cover every formulation of PrEP. Efforts are underway to improve insurance compliance with the ACA preventive services requirements for PrEP in general, but that advocacy faces an uphill road when Gilead is able to set whatever arbitrary starting price point it wants.
Another concern is that as new products, such as lenacapavir, come online, fullcourt marketing efforts may undercut the nascent market for generic oral forms of PrEP, which are safe, effective, and at less than $1 per pill, high quality but dirt cheap. This is exactly what happened when Gilead’s new oral PrEP regimen was approved the year before its first brand-name project went generic.
In the US, newer is often automatically dubbed better. And while the clinical trial results for lenacapavir are certainly impressive, it is not at all clear that the future of PrEP should be to push everyone onto long-acting brand-name products. Given the dismal uptake during these first few years of Apretude, the ViiV long-acting injectable for PrEP, it is also unclear if the market for long-acting products is quite as large as manufacturers say it is. At the end of the day, choice of the PrEP product that works best for the individual is the gold standard. What makes that choice possible is a fair price.
In the face of seemingly intractable drug access challenges in this country, there is still always the potential for an alternative future here. We can envision a scenario in which Gilead—perhaps facing significant pressure from the US advocacy community and the US government—commits to working with existing public health programs and, eventually, a national PrEP program to set a public health price point for lenacapavir to ensure a simple, transparent, and intuitive pathway for uninsured populations. This would be a new, bold approach—not a repeat of past charitable and donation programs that fail to provide an integrated approach to PrEP delivery. Instead, it would be a true, significantly lower price point that empowers public programs and health departments to come up with simple and comprehensive financing and delivery mechanisms that are highly accessible for end users. Only then will we stop repeating the same pattern in which the bang of a big scientific breakthrough is followed by the whimper of inaccessibility.
Credit: healthaffairs.org
5 Ways In Which You Are Harming Your Nervous System And Lowering Stress Response
Team Metabolic Health
Common Lifestyle Mistakes
You may be unknowingly making some common mistakes that may impact your nervous system. It can affect nervous system regulation and also contribute to chronic stress, as opposed to reducing it. It is important to make a few small changes to your routine that can make a big difference. Scroll down to find out about the five simple habits that you need to change.
Checking Your Phone
Avoid checking your phone the moment you wake up. It can trigger a stress response by overwhelming you with notifications in the form of work emails and social media updates, says Marina Wright, a nutritionist.
Not Taking Breaks
You must take breaks from work, because constantly working without a pause can lead to ‘mental fatigue’ and reduced productivity, the nutritionist says, adding that it can keep the nervous system in a state of ‘continuous activation’.
Skipping Meals
According to Wright, skipping meals can cause your blood sugar levels to drop. It can trigger a stress response and disrupt the normal functioning of the nervous system. Skipping a meal can also lead to irritability, anxiety and fatigue.
Spending Time Indoors
It is not wise to spend time indoors all day every day. It can limit your exposure to natural light, which can impact your mood negatively, and also increase stress. When there is a lack of sunlight, your circadian rhythm gets affected and you may feel a drop in energy levels.
Too Much Caffeine
Overconsumption of caffeine can stimulate the nervous system, and not in a good way. According to the expert, too much caffeine can put you in a constant state of alertness that can heighten your stress and anxiety. Consuming it late in the day can also disrupt your sleep.
Credit: thehealthsite.com
Thyroid swelling: What does it indicate? How to get it treated?
Team Metabolic Health
Thyroid is a gland located at the base of the neck responsible for producing thyroxine (T-4) and triiodothyronine (T-3). These hormones help in normal growth and metabolism of the body. Thyroid issues are common in women and can be treated with hormonal supplements. However, swelling of the thyroid gland can indicate other conditions, and should be treated with urgency. In an interview with HT lifestyle, Dr. Satish Nair, academic head and consultant – Skull Base Surgeon and ENT – Head and Neck Surgeon, Manipal Hospital Old Airport Road, Bengaluru, explained thyroid swelling and how it should be treated. “Although thyroid swelling is common among females, men are also affected by it,” said Dr. Satish Nair.
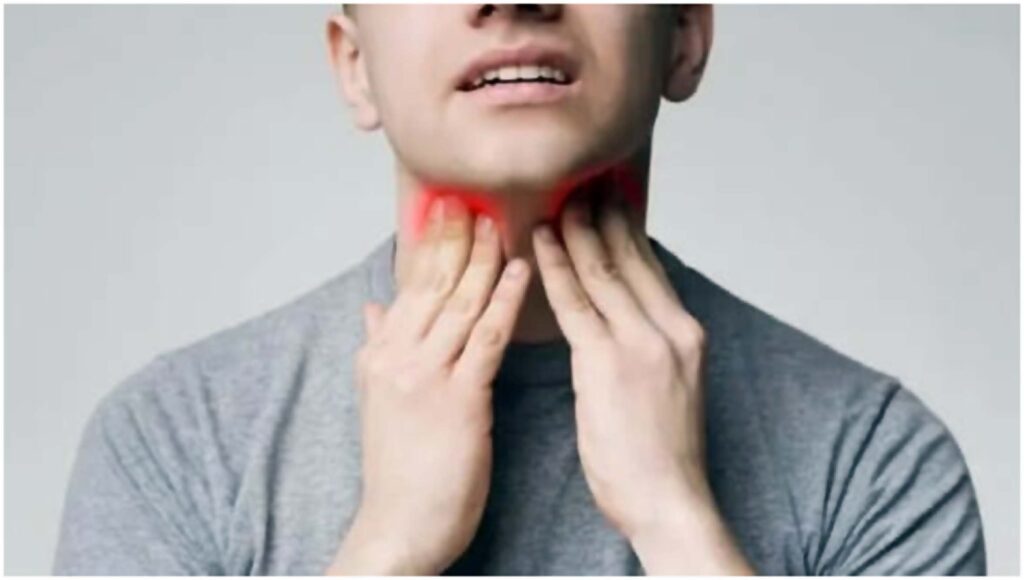
Thyroid swelling is seen as swelling in the front portion of the neck which gradually increases in size. (Credit: Unsplash)
Thyroid swelling: What does it indicate?
“Thyroid swelling is seen as swelling in the front portion of the neck which gradually increases in size. Although thyroid swelling is common among females, men are also affected by it. It can be accompanied by hormonal disturbances too. A swelling in the thyroid can be a simple cyst or nodule or a tumor or cancer in rare cases. 90% of thyroid swelling in women is non-cancerous whereas thyroid swelling in men has a 95% chance of being cancerous,” said Dr. Satish Nair.
Thyroid swelling: Evaluation process
Dr. Satish Nair explained the three-test procedure that is recommended in evaluating thyroid swelling – “When you notice a swelling in the neck that is persistent for more than 3 weeks, it is advisable to consult an ENT / Head and Neck Surgeon. There are three tests that are required for diagnosing thyroid swelling. First is the thyroid hormone test followed by an ultrasound scan of the neck which shows the size, site, and nature of the swelling. The third test you will be advised is a needle aspiration test from the swelling (FNAC – fine needle aspiration cytology) which is done to know the type of cells (cancerous or tumor) in the swelling.”
Thyroid Swelling: Treatment procedures
“Wait and watch is recommended in a female patient with a small swelling of less than 1 cm. Generally, it is advised that a thyroid swelling more than 4 cm in size, swelling in a male, age more than 55 years, and if the tests point towards cancer or swelling is accompanied by pressure symptoms (difficulty in swallowing, breathing, and voice change), it requires surgery as the mainstay of treatment. The decision to remove either partial or complete thyroid gland depends on the test results,” added Dr. Satish Nair.
Disclaimer: This article is for informational purposes only and not a substitute for professional medical advice. Always seek the advice of your doctor with any questions about a medical condition.
Credit: Hindustan Times
Friday Tips! Does A Five Minute Nap Actually Help?
Team Metabolic Health
As a sleep enthusiast, I must confess that there’s nothing quite like the rejuvenating power of a good nap. When that afternoon slump rolls around, I’m always on the lookout for a cozy nook where I can catch a few winks and recharge my batteries.
But in this fast-paced world, we don’t always have the luxury of time to indulge in a full siesta. That’s where the elusive ‘five-minute nap’ comes into play – but does it really work? Can such a brief snooze actually make a difference in our day-to-day lives?
I’ve often found myself wondering about this very question as I lay down for those precious moments of shut-eye. As someone who’d gladly be an ambassador for napping if given the opportunity, it feels almost too good to be true that we could reap any benefits from such a short slumber.
So, let’s dive into the science behind these mini-naps and see if they’re truly worth incorporating into our daily routines!

The Benefits Of Napping
Oh, you must be joking! Who would ever think that a short nap could actually have any benefits?
I mean, it’s not like countless studies and sleep experts have continually proven the positive effects of napping on our mood, focus, and overall well-being. And let’s not even mention those meditation techniques that can work wonders with just a few minutes of practice each day.
But seriously, who needs all that when we can simply push through our days in a state of constant fatigue and mental fog?
As a dedicated sleep enthusiast, I can assure you that napping is no laughing matter. It has been shown to improve memory consolidation, enhance creativity, and even boost your overall cognitive performance.
Plus, did you know that naps are actually an effective way to experience different sleep stages in a short amount of time?
So the next time you find yourself feeling sluggish or unable to concentrate on your tasks, remember that a quick nap might be just what you need to recharge your batteries and get back on track. Don’t underestimate the power of some shut-eye; even five minutes can make all the difference!
What Happens During A Nap?
I’m a huge fan of napping, and I’m so interested in learning more about what’s happening to my body and brain while I’m snoozing! I’m curious to discuss how my bodily reactions and brain activity are impacted by a five minute nap.
Bodily Reactions
Isn’t it amazing how our bodies respond to even the briefest moments of rest?
When we allow ourselves to take a quick nap, the mind-body connection kicks into high gear, rejuvenating us from head to toe.
With cognitive stimulation on pause, our brain takes this opportunity to consolidate memories and refresh neural connections.
Meanwhile, our muscles relax and our heart rate slows down, allowing for a momentary respite from the daily grind.
It’s truly remarkable what just five minutes of shut-eye can do for us – who wouldn’t want to embrace such a simple yet powerful tool in maintaining our overall well-being?
Brain Activity
Now, let’s dive deeper into the fascinating world of brain activity during a nap, shall we?
As a sleep enthusiast, I find it incredibly interesting how mindful sleeping can significantly impact our cognitive abilities.
When we catch some Zzz’s, even for a short while, our brain starts working its magic – cycling through various sleep patterns and allowing us to recharge mentally.
During this time, our mind is busy processing information, sorting memories, and strengthening neural connections – all crucial for optimal functioning.
So next time you’re feeling sluggish or overwhelmed, don’t hesitate to give your brain the much-needed break it deserves with a power nap!
How To Take A Five-Minute Nap
Now that we’ve explored the wonders of what happens during a nap, let’s dive into the magic of five-minute power naps! Though it might seem like a short amount of time, a five-minute nap can actually provide some benefits. Power naps are an excellent way to recharge your brain and boost your productivity throughout the day.
To make the most out of your five-minute nap, try these three simple napping techniques:
- Find a quiet and comfortable place where you can sit or lie down without any distractions.
- Set a timer for exactly five minutes to ensure that you don’t oversleep.
- Close your eyes and take slow, deep breaths as you drift off into a light sleep.
Remember, practice makes perfect! The more you incorporate power naps into your daily routine, the better you’ll become at falling asleep quickly and reaping the rewards of this energizing slumber.
So go ahead – give yourself permission to take a break and indulge in a quick snooze; you deserve it!
The Science Behind Quick Naps
You might be wondering if a quick five-minute nap can really make a difference in your day. Well, let me tell you, as a sleep enthusiast, I’ve always been curious about the science behind quick naps and how they affect our sleep quality. It turns out that short naps, also known as “power naps,” can indeed provide some benefits to our alertness and overall cognitive functioning.
The key to understanding the effectiveness of quick naps lies in our sleep cycles. During the course of a normal night’s sleep, we go through several stages of light and deep sleep, each lasting approximately 90 minutes. When we take a short nap, we’re only able to experience the lighter stages of sleep – but that’s not necessarily a bad thing! In fact, research suggests that even brief periods of rest can help us feel more refreshed and focused. Just take a look at this handy table that highlights the benefits of different nap durations:
| Nap Duration | Benefits |
| 5-15 minutes | Improved alertness & concentration |
| 20-30 minutes | Enhanced creativity & mood |
| 60-90 minutes | Boosted memory & learning |
So while it may seem counterintuitive at first glance, there is indeed some value in taking those ultra-short five-minute naps. They may not offer the same rejuvenating effects as longer snoozes, but when time is limited or you just need a quick pick-me-up during your busy schedule, don’t hesitate to close your eyes for those precious few moments. You’ll likely find yourself feeling more awake and ready to tackle whatever comes your way!
Is A Five-Minute Nap Worth It?
So, you might be wondering, is a five-minute nap even worth it?
Well, as a sleep enthusiast, I can tell you that it certainly has its merits! Although it may not be as restorative as a longer snooze, there are some short-term effects of taking such a brief nap that can be beneficial.
For one thing, it can provide a quick energy boost and help to improve your mood. Additionally, it can also help with enhancing focus and alertness in the moments immediately following the nap.
Now, when considering the long-term impacts of consistently taking five-minute naps, there’s not much evidence to suggest that they’ll have any significant effect on your overall sleep health.
However, if these brief naps are helping you feel more energized and focused throughout your day without disrupting your regular sleep schedule at night, then why not give them a try?
Just remember that everyone’s sleep needs are different, so it’s essential to listen to your body and adjust accordingly.
Conclusion:
So, does a quick five-minute nap truly make a difference? As a sleep enthusiast, I must say that even the shortest of naps can provide an energy boost and increase alertness. However, it’s essential to remember that while we may enjoy the benefits of a power nap, it shouldn’t replace proper sleep.
In my experience, finding the perfect balance between work, life, and rest is crucial. Sure, we might not always have time for a full siesta but squeezing in a brief five-minute nap can help us reset and recharge. We cannot forget the importance of longer naps or quality nighttime sleep for overall health and well-being.
In conclusion, as someone who loves their sleep, I encourage everyone to embrace the power of napping – even if it’s just for five minutes! The key is to find what works best for you and make sure you prioritize rest when needed.
Remember, every little bit counts when it comes to catching those precious Zzzs!
Credit: https://tiptopsleep.com/
Lilly releases Zepbound® (tirzepatide) single-dose vials, expanding supply and access for adults living with obesity
Team Metabolic Health
Eli Lilly and Company (NYSE: LLY) has announced Zepbound (tirzepatide) 2.5 mg and 5 mg single-dose vials are available for self-pay for patients with an on-label prescription, significantly expanding the supply of Zepbound in response to high demand. The single-dose vials are priced at a 50% or greater discount compared to the list price of all other incretin (GLP-1) medicines for obesity. This new option helps millions of adults with obesity access the medicine they need, including those not eligible for the Zepbound savings card program, those without employer coverage, and those who need to self-pay outside of insurance.
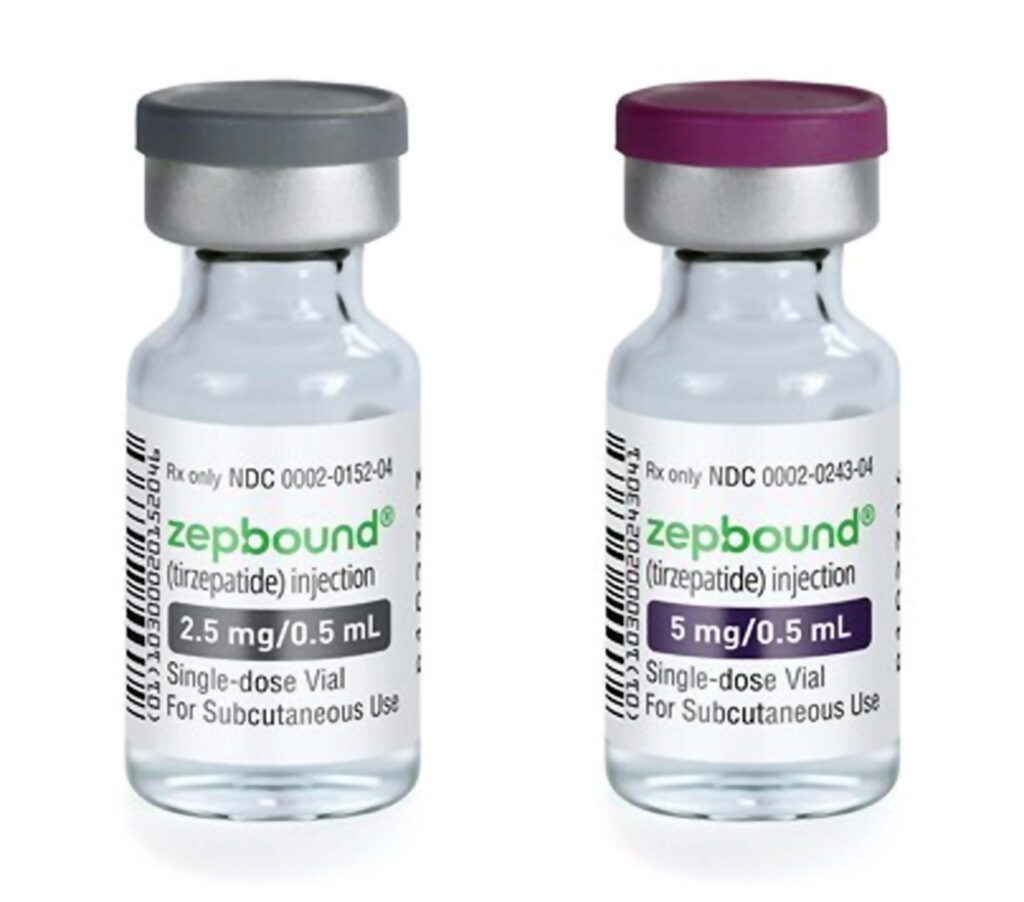
“We are excited to share that the Zepbound single-dose vials are now here, further delivering on our promise to increase supply of Zepbound in the U.S.,” said Patrik Jonsson, executive vice president, and president of Lilly Cardiometabolic Health and Lilly USA. “These new vials not only help us meet the high demand for our obesity medicine, but also broaden access for patients seeking a safe and effective treatment option. In a clinical study, the 5 mg maintenance dose helped patients achieve an average of 15% weight loss after 72 weeks of treatment and has been a powerful tool for millions of people with obesity looking to lose weight and keep it off.”
Lilly has created a new self-pay pharmacy component of LillyDirect where patients with a valid, on-label prescription from the health care provider of their choice can purchase the vials. Distributing the vials via this channel ensures patients and providers can trust they are receiving genuine Lilly medicine, building on the company’s efforts to help protect the public from the dangers posed by the proliferation of counterfeit, fake, unsafe or untested knock-offs of Lilly’s medications. Lilly has also taken a vocal stance against the use of obesity medicine for cosmetic weight loss; a multi-step verification process will help ensure the vials are dispensed only to patients who have a valid, on-label electronic prescription from their health care provider. Patients can also purchase ancillary supplies, like syringes and needles, and will have access to important patient-friendly instructional materials on correctly administering the medicine via needle and syringe.
“People living with obesity have long been denied access to the essential treatment and care needed to manage this serious chronic disease,” said James Zervos, chief operating officer, Obesity Action Coalition. “Expanding coverage and affordability of treatments is vital to people living with obesity. We commend Lilly for their leadership in offering an innovative solution that brings us closer to making equitable care a reality. Now, it’s time for policymakers, employers and insurers to work with pharmaceutical companies to ensure no one is left behind in receiving the care they deserve and need.”
A four-week supply of the 2.5 mg Zepbound single-dose vial is $399 ($99.75 per vial), and a four-week supply of the 5 mg dose is $549 ($137.25 per vial) – less than half the list price of other incretin medicines for obesity and in line with the Zepbound savings program for non-covered individuals. The self-pay channel enables a transparent price by removing third-party supply chain entities and allowing patients to access savings directly outside of insurance.
“Despite obesity being recognized as a serious chronic illness with long-term consequences, it’s often misclassified as a lifestyle choice, resulting in many employers and the federal government excluding medications like Zepbound from insurance coverage,” said Jonsson. “Outdated policies and lack of coverage for obesity medications create an urgent need for more innovative solutions. Bringing Zepbound single-dose vials to patients will help more people living with obesity manage this chronic condition. We will also continue to advocate for a system that better aligns with the science.”
In a clinical study, tirzepatide 5 mg, along with a reduced calorie diet and increased physical activity, achieved an average of 15% weight loss over 72 weeks compared to 3.1% for placebo. Zepbound is the first and only obesity treatment of its kind that activates both GIP (glucose-dependent insulinotropic polypeptide) and GLP-1 (glucagon-like peptide-1) hormone receptors. Zepbound tackles an underlying cause of excess weight. It reduces appetite and how much you eat. Zepbound is indicated for adults with obesity, or those who are overweight and also have weight-related medical problems, to lose weight and keep it off. Zepbound should be used with a reduced-calorie diet and increased physical activity. It should not be used in children under 18 years of age or with other tirzepatide-containing products or any GLP-1 receptor agonist medicines. Zepbound has not been studied in patients with a history of pancreatitis, or with severe gastrointestinal disease, including severe gastroparesis, and it is unknown if patients with a history of pancreatitis are at higher risk for developing pancreatitis on Zepbound.
Zepbound is also available in 2.5 mg, 5 mg, 7.5 mg, 10 mg, 12.5 mg, or 15 mg per 0.5 ml doses in a single-dose pen (autoinjector). The recommended maintenance dosages are 5 mg, 10 mg, or 15 mg injected subcutaneously once weekly.
Credit: www.lilly.com
Eating Meat Is Linked With Diabetes Risk, New Studies Suggest
Team Metabolic Health
For sausage, salami and steak lovers, the news has not been good. Scientists have been consistently finding links between red and processed meat consumption and heart disease, some types of cancer and earlier death.
And now, two recent studies have added to the growing body of evidence that a meat-heavy diet may increase the risk of Type 2 diabetes.
In one of the studies, published today in The Lancet Diabetes and Endocrinology, researchers analyzed data from nearly two million adults participating in 31 studies across 20 countries, including the United States and parts of Europe and Asia.
The researchers reviewed survey data on participants’ diets and then looked at their health an average of 10 years later. After adjusting for other risk factors like smoking, a higher body mass index, physical inactivity and a family history of diabetes, they found that for every 1.8 ounces of processed meat the participants ate each day, their risk for Type 2 diabetes increased by 15 percent. (This is equivalent to a medium-sized sausage or two to three slices of bacon.) For every 3.5 ounces of unprocessed red meat they consumed daily, their risk increased by 10 percent.
The data also suggested that one serving of poultry per day was associated with an 8 percent increase in Type 2 diabetes risk, but this finding was less consistent and only significant in the European studies, so more research is needed, said Dr. Nita Forouhi, a professor of population health and nutrition at the University of Cambridge who led the study.
The takeaway, she said, is that the less red and processed meat you eat, the better.

Credit: Getty Images
Why Eating Meat Might Increase Your Risk
These findings jibe with previous research, including a large U.S. study published in October.
There are several potential explanations for why regular meat consumption might increase Type 2 diabetes risk, Dr. Forouhi said.
One is that red and processed meats tend to be higher in saturated fats than in unsaturated fats, a composition associated with greater insulin resistance, which can lead to Type 2 diabetes.
People who eat more meat may also eat fewer healthful foods, like fruits and vegetables, Dr. Forouhi added.
Cooking meat at high temperatures, such as through pan frying or grilling over an open flame, can also form certain compounds that may cause cell damage, inflammation and insulin resistance — all of which can contribute to Type 2 diabetes.
A study published last week in the journal Nature Metabolism added evidence for another longstanding hypothesis: that heme iron — a type of iron found in high levels in red meat (and to a lesser extent in fish and poultry) — may contribute to Type 2 diabetes.
The researchers followed nearly 205,000 predominantly white U.S. adults for up to 36 years, during which time about 21,000 of them developed Type 2 diabetes.
Those with the highest heme iron consumption — mainly coming from eight to 10 servings of unprocessed red meat per week — were 26 percent more likely to develop Type 2 diabetes than those with the lowest consumption. They also had higher lipid levels, markers of insulin resistance and inflammation, and other compounds linked with Type 2 diabetes in their blood, the study revealed.
This added to existing evidence suggesting that heme iron is likely an important player in the link between red meat and Type 2 diabetes, said Dr. Frank B. Hu, a professor of nutrition and epidemiology at the Harvard T.H. Chan School of Public Health who led the study.
On the other hand, heme iron did not explain the link between processed red meat and Type 2 diabetes, Dr. Hu said. Other components, such as their preservative and sodium levels, are probably more important to the risk of processed meats, he added.
Iron is an essential nutrient, but too much may cause inflammation and damage the pancreas, increasing diabetes risk, said Dr. Dariush Mozaffarian, a cardiologist and professor of medicine at Tufts University, who was not involved with the study.
How Much is Too Much?
While regular consumption of both red and processed meats have been associated with poorer health, processed meats have the strongest and most consistent link with Type 2 diabetes and other conditions, Dr. Mozaffarian said. This is clearly a “class to avoid,” he added.
Processed meats include not only stereotypically unhealthy products like hot dogs and bacon, but also potentially healthy-seeming ones like turkey sausages and deli meats.
This doesn’t mean you should never eat these foods, Dr. Mozaffarian said. Instead, think of them as occasional treats, he added.
As for red meat, there’s not enough data to say exactly how much is too much, Dr. Hu said, but based on the current evidence, a serving or two per week — like a small pork chop or a couple meatballs over spaghetti — is probably OK.
Dr. Hu recommended shifting from a “meat centric” diet to one that incorporates plenty of plant-based foods like whole grains, fruits, vegetables and legumes — all of which have been linked with a lower risk of Type 2 diabetes.
Credit: The New York Times
New obesity treatments could speed up metabolism, finds clinical trial
Team Metabolic Health
Weight-loss medications such as Ozempic, Wegovy or Monjaro may not just suppress appetite but could also speed up metabolism. This is according to a (opens in a new window)new study carried out at St Vincent’s University Hospital (SVUH).
A randomized controlled trial led by Professor Donal O’Shea, SVUH and UCD School of Medicine, found that there was a strong relationship between the amount of weight lost and increases in metabolic activity caused by daily use of medications based on the hormone Glucagon-like peptide-1 (GLP-1).
Ozempic is a diabetes medicine that in recent years has become popular as a new an anti-obesity medication Credit: Chemisy4U
The findings published in the Journal of the Obesity Society challenges the belief that recent weight loss medications work just by simply causing those who use them to eat less.
“It always seemed oversimplistic to me that these new treatments were just making people eat less,” said Professor O’Shea.
“So, this study finding is an exciting step forward in our understanding of how these new medicines for obesity work.”
Adding: “This study challenges the main narrative about these newer treatments which is that they simply make you eat less, and that any action on energy burn is minimal.
“The findings also provide science to support the fact that the treatment of obesity is not simply to eat less and move more – that’s the prevention piece – treatment is more complex than that.”
In the randomised controlled trial, which involved 30 patients, those who had low metabolic activity before starting treatment benefited the most from medications such as Ozempic, Wegovy and Monjaro.
The patients had specialised imaging of the fat within their abdomen using a PET-CT scanner, with scans carried out before and after six months of treatment.
The results were showed a strong correlation between the increase in metabolic activity and the degree of weight loss. Furthermore, those with low metabolic activity before starting treatment benefited the most from it.
“The strength of the association is surprising given the relatively small numbers studied and suggests this increase in metabolic activity is a significant contributor to how these drugs work,” said Professor O’Shea.
“Safe medical treatment for obesity is still in its infancy and we need to understand fully how the treatment works. Understanding how these agents increase energy burn should be an important part of future research.
“I hope the companies involved in the development of these treatments will examine this area in more detail because these are very expensive studies to carry out and we are very grateful to the Health Research Board and University College Dublin for supporting it.”
The study ‘(opens in a new window) GLP‐1 therapy increases visceral adipose tissue metabolic activity: lessons from a randomized controlled trial in obstructive sleep apnea’ was co-authored by (opens in a new window) Professor Silke Ryan, SVUH, funded by the Health Research Board and supported by University College Dublin.
Credit: University College Dublin
Forget BMI—Here’s Why You Should Focus on “Metabolic Health”
Team Metabolic Health
We’ve all heard of the metabolism: how well—or poorly—your body processes energy. Metabolism is often talked about in terms of weight loss or gain, but fundamentally, metabolism is about “converting the potential energy in food to a usable form of energy that powers our cells,” says Casey Means, MD, best-selling author of Good Energy: The Surprising Connection Between Metabolism and Limitless Health and a co-founder of Levels, a metabolic health tracking device. Your metabolism dictates a lot of things (weight included, but not exclusively), and when it’s unhealthy, it can lead to a range of diseases from type 2 diabetes to Alzheimers to behavioral disorders to cardiovascular issues to autoimmune diseases.
Metabolic health, on the other hand, is about far more than your BMI—which is an outdated metric. It refers to the optimal functioning of the entire metabolic system, including blood glucose, blood pressure, blood lipids, history of cardiovascular disease, and adiposity. According to a recent survey, 93.2 percent of Americans aren’t metabolically healthy.
This term is not really in our everyday vocabulary and is even relatively new to the medical field—partly because it requires a holistic look at our bodies, which Means says is lacking in our current healthcare system. “There’s this interesting phenomenon happening where the more we actually spend on healthcare in the United States—now 4.5 trillion dollars per year—the sicker Americans are getting,” she says. “It’s much more profitable to wait for people to get sick and then treat their various symptoms and conditions with whack-a mole medicine and chronic disease management for life.”
But failing to take a holistic approach is impacting people’s quality of life. “Metabolic health controls everything,” says Dr. Markyia Nichols, MD, chief medical officer at Ciba Health, a root-caused healthcare organization. “It’s really maintaining the overall balance; it affects every system of the body.” When metabolic health is optimized, people often experience increased energy, improved mental clarity, better physical performance, and even an increased feeling of well-being. Conversely, poor metabolic health can contribute to chronic fatigue and brain fog, making it harder to engage in everyday activities and fully enjoy life. Plus, “if you get your metabolic health under better control, it’s slashing the risk of almost every chronic symptom and disease,” says Means.
Now, medical providers have more tools at their disposal to get a comprehensive look at your metabolic health through a detailed lifestyle questionnaire, blood glucose monitoring, wearable data tracking, blood work panels, blood pressure testing, and weight circumference measurements. These assessments help identify underlying issues that might not be apparent through traditional health metrics like weight or BMI alone. If testing comes back outside of optimal ranges, Nichols and her team would dive in to find the underlying triggers and influences based on lifestyle. “We’re not just treating one symptom, we’re really looking at it all,” says Nichols. Even if you can’t see a root-cause or preventative healthcare provider, you can get most of these lab tests covered by insurance—you just may need to be proactive and ask your doctor.
If you’re not already metabolically healthy, not all is lost: You don’t just naturally have “fast” or “slow” metabolism, as we once assumed. “The empowering part,” says Means, “is that there is so much we can do to improve our mitochondrial function.” According to Michal Mor, cofounder of Lumen, a metabolic tracking device, we can influence our metabolic health by prioritizing four lifestyle categories: Nutrition, movement, sleep, and stress levels.
Ultimately, the most powerful things you can do to optimize your metabolic health aren’t novel—they’re pretty on par with recommendations from nearly every health and longevity expert. Get enough sleep, eat a nutrient-rich diet of real foods, incorporate daily movement (with at least some of it strenuous), and keep those damn stress levels under control.
Credit: GQ
What Is ‘Smart’ Insulin? Can It Cure Type 1 Diabetes? How Will It Control Blood Sugar Levels In Real Time?
Team Metabolic Health
The new glucose-responsive insulins promise to end constant glucose monitoring, and could allow patients to swallow a pill or inject insulin once every morning.
People with type 1 diabetes will in future need to take insulin once a week, say experts… The effect of new glucose-responsive insulins would be similar to that produced by a functioning pancreas, which only releases insulin when it is needed in the body
“The funded six new research projects address major shortcomings in insulin therapy. Potentially minimising the risk of hypoglycaemia through an insulin-glucagon combination would ease one of the major concerns associated with insulin therapy today. Therefore, these research projects, if successful might do no less than heralding a new era in insulin therapy,” Tim Heise, Vice Chair of Novel Insulins Scientific Advisory Panel of the Type 1 Diabetes Grand Challenge, said.
What is Type 1 Diabetes?
Blood sugars, also called blood glucose, are the primary source of energy in the body. When you eat food, the body breaks down most of it into glucose, which is released into the bloodstream. When glucose levels go up, the pancreas releases the hormone insulin, which helps glucose to be used as energy by your cells.
Those with type 1 diabetes have a pancreas that either cannot produce insulin or produces very little of it. High levels of glucose can lead to heart disease, kidney problems, extreme fatigue and other serious illnesses.
What is the Difference Between Type 1 and Type 2 Diabetes?
Type 1 diabetes is an autoimmune disease in which the body cannot make any insulin at all. The insulin-producing cells have been attacked and destroyed by the body’s immune system. While in type 2 diabetes, the body does not make enough insulin.
Type 1 diabetes affects 8% of people living with diabetes and type 2 diabetes affects 90% of people living with diabetes.
The risk of developing type 1 diabetes depends on family history and genes, and is often diagnosed in childhood or when you are under 40.
Type 2 diabetes develops with age, and your ethnicity can increase your risk. For instance, if you are white and over 40, the risk of developing type 2 diabetes is higher, whereas in African-Caribbean, Black African, Chinese or South Asians, chances of developing the illness increase if you are over 25.
In India, there are around 8.6 lakh people with Type 1 diabetes, with one in six young people dying without a diagnosis. An estimated 77 million people above the age of 18 years are suffering from type 2 diabetes, and nearly 25 million are prediabetics (at a higher risk of developing diabetes in near future), according to the World Health Organization.
The 10 countries with the highest estimated prevalence — USA, India, Brazil, China, Germany, UK, Russia, Canada, Saudi Arabia and Spain — account for 5.08 million or 60% of global cases of Type 1 diabetes.
What is the Need for a Better Insulin?
The glucose levels in the body constantly change depending on your stress levels, whether you have exercised, the foods you eat and the hormone levels. This makes it hard for the people with type 1 diabetes to maintain a stable blood sugar level, even with the latest technology to administer insulin.
A standard insulin can stabilise blood sugar levels for a certain time, but it cannot typically help with future fluctuations. It means patients often need to inject more insulin again within just a few hours.
Faster insulins are also needed to improve the function of insulin pumps and hybrid closed loop technology – a system that relies on the stored insulin responding in real-time to changing blood sugar levels.
What is a Smart Insulin?
The new glucose-responsive insulins (GRIs) become active when they detect a change in sugar levels in the blood to prevent hyperglycaemia (high blood glucose). They become inactive again when levels drop below a certain point, avoiding hypoglycaemia (low blood glucose). In future, patients may only need insulin once a week, experts believe.
The effect of a smart insulin would be similar to that produced by a functioning pancreas, which only releases insulin when it is needed in the body.
The GRIs promise to end constant glucose monitoring, and could allow patients to swallow a pill or inject insulin once every morning.
What are the 6 Research Projects for Smart Insulins?
The six projects include teams at Stanford University in the US, Monash University in Australia and Zhejiang University in China. The aim is to accelerate development and launch trials as soon as possible.
The Monash University project involves development of a second generation of nano sugar-insulin system, based on advanced nanotechnology. These nano sugars react to very small changes in blood glucose and release insulin only when glucose levels are outside a range, without any intervention from the patient.
Researchers at the Wayne University are working to develop a “smart insulin” which can detect changes in blood glucose levels and respond by releasing the right amount of insulin at the right time.
The third project, conducted by researchers at the Jinhua Institute of Zhejiang University in China, involves novel insulins that respond immediately to rising blood glucose levels.
At the University of Notre Dame, US, researchers developed a smart insulin delivery system that uses tiny particles called nanocomplexes, which contain insulin. These nanocomplexes can also be injected under the skin to create a reservoir to automatically release insulin if blood sugar rises.
Scientists at the Stanford University, US are working on developing and testing an ultrafast-acting insulin that’s only active when needed and could reduce the risk of blood glucose highs and lows in people with type 1 diabetes.
A team of researchers at the Indiana University, US will combine insulin and glucagon in their project, to prevent the highs and lows in blood glucose.
Dr Elizabeth Robertson, the director of research at Diabetes UK, said, as quoted by The Guardian, the projects had the potential to revolutionise type 1 diabetes treatment. “By supporting these groundbreaking research projects, we are aiming to develop new insulins that more closely mimic the body’s natural responses to changing blood sugar levels.
Credit: News18
